Archive : Article / Volume 1, Issue 1
- Research Article | DOI:
- https://doi.org/10.58489/2836-3590/002
Determination of Heavy Metals Pollution in Soil and Tree Rings along Haatso- Atomic Road in Ghana
1Department of Biological, Physical & Mathematical Sciences, University of Environment &Sustainable Development, Somanya, Ghana
2Department of Physics, College of Science, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
3Department of Pharmaceutical Sciences, Sunyani Technical University, Sunyani, Ghana
4Department of Physics, University of Cape Coast, Ghana
5Graduate School of Nuclear and Allied Sciences, Ghana Atomic Energy Commission, University of Ghana
6Department of Basic Science, University of Health and Allied Sciences, Ho, Ghana
George Edusei, Department of Biological, Physical & Mathematical Sciences, University of Environment & Sustainable Development, Somanya, Ghana
dusei G, Subaar C, Gyan E, Osei-Owusu J, Edziah R, Dofuor AK, Tandoh JB, Kwakye R, Ntim E. Determination of Heavy Metals Pollution in Soil and Tree Rings along Haatso-Atomic Road in Ghana. J. Pollut. Eff. Community Health. Vol. 1 Iss. 1 (2022). DOI: 10.58489/2836-3590/002
© 2022 George. E, this is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Received Date: 01-08-2022
- Accepted Date: 11-08-2022
- Published Date: 19-08-2022
Heavy metal contamination, Enrichment factors, Geo-accumulation index, Roadside soil, Human health risk
Abstract
Environmental pollution caused by heavy metals from automobiles has received much attention in recent years. In this study, heavy metal pollutants in soil and tree rings were determined along the Haatso-Atomic Road in the Greater Accra region of Ghana using X-ray fluorescence. Contamination factor, enrichment factors, geo-accumulation index and health risk assessment model were used for assessing the contamination level of heavy metals. X-ray fluorescence (XRF) was used to investigate the presence of the following heavy metals: Cu, Mn, Zn, Pb, Cd, Fe and Ni. The data was analyzed using Pearson’s coefficient of correlation. Correlation analysis showed that the origin of heavy metals in soil and tree rings are concentrations of Cu, Mn and Cd increase in soil and tree ring as this can be attributed to vehicle exhausts and abrasion of vehicle parts (such as tyres) produced in traffic. According to I-geo’s findings, Cd was determined to be unpolluted, while Cu, Mn, Zn, Pb, Fe, and Ni were found to be moderately contaminated. The Haatso-Atomic Road’s soil chemical component enrichment factor data show that anthropogenic elements (such as Zn, Cu, and Pb) were moderately to significantly enriched in comparison to crustal elements. With regards to health risk, the results of this study indicate that non-carcinogenic risks caused by heavy metals were insignificant while the carcinogenic risk caused by Cd was found to be significant. The outcomes of this study will help to guide national policy in the areas of vehicular heavy metal pollution and other anthropogenic causes of pollution.
Introduction
Pollution of the natural environment by heavy metals is a general problem as these metals are nondegradable and as such most of them have adverse effects on living organisms including humans, and the environment when acceptable concentration levels are exceeded (Censi et al, 2006). Heavy metals frequently reported in literature with regards to potential hazards and occurrences in contaminated soils are Cd, Cr, Pb, Zn, Fe and Cu (Akoto et al., 2008). The adverse effects of heavy and trace elements in the soil system are increasingly becoming a problem of global concern since soil constitutes a central component of rural and urban environments (USDA, 2001). The sources of these contaminants mainly include natural occurrences derived from parent materials and human activities.
Anthropogenic inputs originate from industrialization in the form of atmospheric deposition, waste disposal, waste incineration, urban effluents, traffic emissions, fertilizer application and long-term application of wastewater in agricultural lands (Bilos et al., 2001; Koch & Rotard, 2001). Various researchers have found that the concentrations of the metals such as Pb, Cu, Zn, Cd and Ni decrease rapidly within 10 to 50 m from the roadsides (Joshi et al., 2010; Pagotto et al., 2001). Heavy metals may enter the human body through dust inhalation, direct ingestion of soil and consumption of food plants grown in metal contaminated soils (Cambra, 1999).
A number of studies have shown the ability of trees to take up and incorporate pollutants into their annual growth rings (Nabais et al., 2001; Speer, 2010). Indeed, tree-rings have been used to provide annual records of pollution over decades, tracing pollutants on a spatial and temporal scale in relation to their sources (Cocozza et al., 2016; Danek et al., 2015; Odabasi et al., 2015). In the last decades, many studies were carried out to reconstruct the temporal and spatial patterns of heavy metals recorded in tree rings of different species (Alahabadi et al., 2017; Watmough ,1997).
Haatso-Atomic in the Greater Accra region of Ghana has undergone rapid urbanization over the past several decades. However, increasing emissions of heavy metals associated with a fast-growing population and industrial activities has put huge pressure on the environment and further irritated ecosystem function. Accompanied with increasing anthropogenic activities, and the demands for a better environment, the levels of health risk exposure to heavy metals in Haatso-Atomic are of concern. In this study, geo-accumulation index and enrichment factor were employed to evaluate the contamination levels of heavy metals in Haatso-Atomic, and the concentrations of heavy metals in tree-ring were tested as a bio-indicator of soil heavy metals. The study specifically focused on identification of possible sources of heavy metals at Haatso-Atomic Road. Therefore, concentrations of heavy metals in tree-rings along the Haatso-Atomic Road were examined to correlate heavy metals temporal change and heavy metal soil contamination.
Material and Methods
Study area
Haatso is a town located in the Ga East Municipal district of the Greater Accra Region of Ghana. It is located on the coordinate (5° 40′ 9.7″ N, 0° 12′ 27″ W) with a population of 19,379 people (CIA, 2021). The Haatso-Atomic Road is adjacent to the Ghana Atomic Energy Commission (GAEC). All samples analyzed for this study were taken from Haasto- Atomic. The road side vegetation at the study sites are dominated by Azadirachta indica (Neem tree) and Swietenia mahagoni (Mahogany tree). These areas are well suited for the study of pollution effects because of the persistent vehicular traffic and other human activities. Figure 1 shows the map of Haatso-Atomic Road with sampling locations indicated in red circles along the main road. All soils were sampled at the surface (15 to 30 cm in depth) using hand-driven stainless-steel augers.
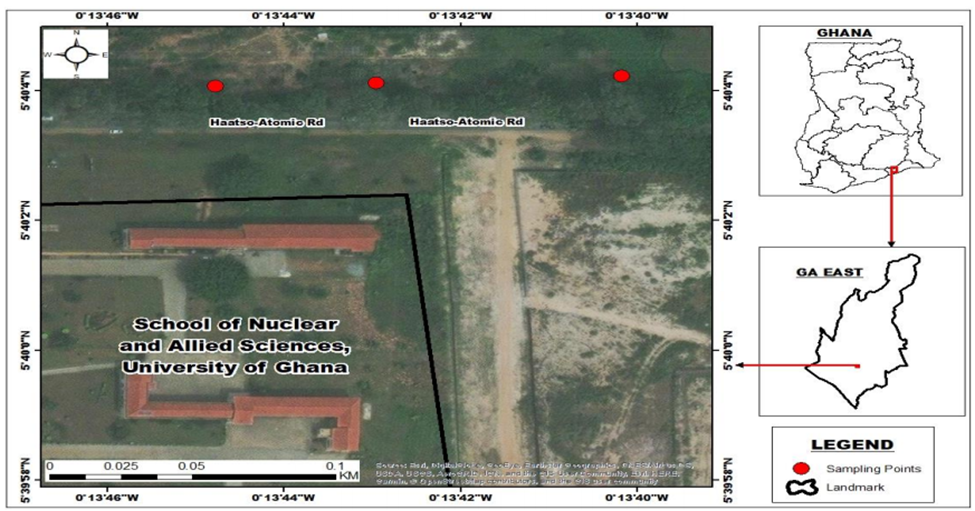
Figure1: Map showing roadside air quality monitoring sites (sampling sites are shown in red circles)
Sample Preparation and Analysis
The soil samples were obtained with a hand auger from the topsoil between depths of 15−30 cm. The first 15 cm of the top layer was not sampled to avoid surface contaminants. Soil samples were collected with a polyethylene scoop and stored in plastic bags. They were then air-dried and passed through a 2 mm plastic sieve to remove gravel and rocks and kept in plastic bags. Briefly, 0.1 g of each homogenized sample was digested by three-acid attack (HF/HClO4/HNO3) in a Teflon vessel and was heated in a microwave oven at 180 °C for 10 min. The digested solution was diluted to a known volume with double distilled water, and was analyzed for metals by XRF. Precision was achieved by triplicate analyses of the same sample and standards.
Swietenia mahagoni (mahogany), which is about one meter (1 m) from the main Haatso-Atomic Road was logged and cross-sections taken for tree ring analysis. Cross-sections of tree samples were dried in the laboratory under supervision. The total length of each radial subsection from the pith to the cortex was measured to the nearest millimeter. Radial sections of the trees with clear growth ring patterns were electronically scanned, counted and growth widths estimated using image J software. Annual rings cut from the radial cross-sections were analyzed for heavy metal composition using X-ray fluorescence analysis.
Statistical Analysis
Correlation analysis was conducted using the SPSS 15 (version 15) statistical package. I-geo and enrichment factor were used to identify the association between heavy metals and evaluate the soil pollution.
Assessment Methods
Contamination factor
The level of contamination of soil by metal is expressed in terms of a contamination factor (CF), the CF is the ratio obtained by dividing the concentration of each metal (CSample) in the soil by the baseline or background value (CBackground) and is calculated as:
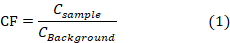
where CF1 refers to low contamination; 1 CF3 means moderate contamination; 3 CF6 indicates considerable contamination and CF6 indicates very high contamination.
Geoaccumulation Index
Geoaccumulation index (I-geo) is an environmental index that enables the assessment of contamination by comparing with current and preindustrial concentrations and has been successfully applied to measurement of soil contamination (Barbieri, 2016; Ololade, 2014). It can be computed using equation 2 (Muller, 1969)
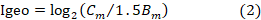
where Cm is the concentration of element (m) recorded in the sediments of the study area, Bm is the geochemical reference value. We used the background values of the study provided by the Environmental Protection Agency of Ghana and 1.5 is the is the background matrix correction due to lithogenic effects, that is, natural fluctuations and anthropogenic influences (Aiman et al., 2016). Table 1 shows the classification for the geoaccumulation index.
| Value | Class | Soil quality |
| Igeo 0 | 0 | Practically uncontaminated |
| 0 Igeo 1 | 1 | Uncontaminated to moderately |
| 1 Igeo 2 | 2 | Moderately contaminated |
| 2 Igeo 3 | 3 | Moderately to heavily contami- nated |
| 3 Igeo 4 | 4 | Heavily contaminated |
| 4 Igeo 5 | 5 | Heavily to extremely contami- nated |
| 5 Igeo | 6 | extremely contaminated |
Table1: Classification for Geo-accumulation index (I-geo)
Enrichment Factor
Enrichment factor (EF) model helps to differentiate between elements originating from anthropogenic activities and those from natural sources. It is used mainly to provide an initial assessment of the degree of contributions from man-made activities to that of the measured atmospheric elemental concentrations. EF compares the ratios for various elements in measured PM concentrations to the corresponding ratios in geological material as a means of confirming the man-made and natural contributions (Chao & Wong, 2002; Cao et al., 2003; Zhang et al., 2008). The EF for each element at any sampling sites was calculated using the Equation 3
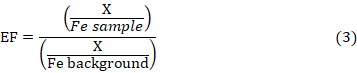
X/Fe-sample is the ratio of heavy metal to Fe concentration in the sample and X/Fe-background is the natural background value for the ratio of heavy metal to Fe
In this study, Fe was used as reference, and the upper continental compositions of the earth’s crust were taken from Taylor and McLennan (1985). Iron (Fe) is most commonly used as reference element because it is an abundant natural content of the soil, possesses good chemical stability and less affected by anthropogenic pollution (Basha et al., 2010). By definition, an enrichment factor closes to unity (EF ~ 1) indicates that the element considered did originate from the soil (Chiarenzelli et al., 2001). The EF of an element is usually taken as being from a natural source if EF < 10. However, if EF > 10 then it is considered to indicate that a significant fraction of the element is contributed from anthropogenic source (Braga et al., 2005). EF values >100 and 1000 are taken as highly and heavily enriched respectively.
Health risk assessment
To assess the potential health risks from the intake of heavy metals through inhalation (Kaur et al., 2020). Human health risks are classified into non-carcinogenic and carcinogenic risks (Adimalla et al., 2020). The average daily dose of carcinogenic (adult) and non-carcinogenic (adult and children) heavy metal, caused by the exposure pathways, was calculated using equation 4 (Fan et al., 2019).

where C is the concentration of heavy metal in soil (mg/kg); Inh is the inhalation rate (m3/d); EF is the exposure frequency (d/a); ED is the exposure duration (a); AT is the time period over which the dose is averaged (d), and it is derived by using pathway-specific period of exposure for non-carcinogenic effects (ED × 365 days/year) and 74.8-year life-time for carcinogenic effects (74.8 years × 365 days/year) averaging time; BW is the average body weight (kg); PEF is the particle emission factor (m3/kg). The values used in these equations are listed in Table 2.
| Exposure Pathway | Parameter | CF | EF | ED | ATcarcinogenic | Non-AT carcinogenic | BW | PEF |
| Inhalation | Adult | 10-6 | 350 | 24 | 74.8 x 365 | ED x 365 | 60.6 | 1.36×109 |
| Child | 10-6 | 350 | 6 | 74.8 x 365 | ED x 365 | 26.5 | 1.36×109 |
Table2: Health risk parameter for heavy metals in soil (Li et al., 2018; Fan et al., 2019; Cao et al., 2020)
The average lifetime daily dose of carcinogenic (children) heavy metal, caused by the exposure pathways, was calculated in equation 5 (Fan et al., 2019).
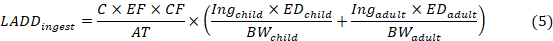
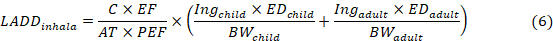
where the definitions of C, CF, EF, AT, Ing, ED, BW, PEF are the same as in Eq. (4)
The assessment of carcinogenic risk was conducted using the following formula (Cui et al., 2018; Fan et al., 2019; Md et al., 2020).
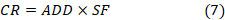
The assessment of non-carcinogenic risk (HQ) was performed using the equation 8 (Cui et al., 2018; Fan et al., 2019; Md et al., 2020).

where ADD is the average daily intake (mg/(kg-day)); SF (dimensionless) is the carcinogenic slope factor (per mg/kg-day); CR is the carcinogenic risk index (Li et al., 2018); RfD is the reference dose (mg/kg-day) (Table 3); and HQ is the non-carcinogenic risk index (Li et al., 2018).
| RfD | SF | |||
| Ele- ment | Ingestion | Inhalation | Inges- tion | Inhala- tion |
| Cu | 4.00×10-2 | 4.02×10-2 | ||
| Zn | 3.00×10-1 | 3.00×10-1 | ||
| Pb | 3.50×10-3 | 3.52×10-3 | ||
| Cd | 1.00×10-3 | 1.00×10-3 | 6.3 | |
| Ni | 2.00×10-2 | 2.06×10-2 | 0.84 | |
| Hg | 3.00×10-4 | 8.57×10-5 | ||
| As | 3.00×10-4 | 3.00×10-4 | 1.5 | 15.1 |
| Cr | 3.00×10-3 | 2.86×10-3 | 42 | |
Table3: RfD and SF of heavy metals in soils with different exposures (Li et al., 2018; Fan et al., 2019; Cao et al., 2020)
Therefore, a study on human health risk assessment of heavy metals has a great significance to the public. According to USEPA (1998), USEPA. (2001), the HQ had a threshold of 1 in
exposure pathways. If HQ<1, there is a small or negligible risk; if HQ>1, there is a non-carcinogenic risk. We calculated human health risk index for children and adults under the exposure pathways according to the exposure doses in Table 3 using the equations (7) and (8).
Results
Temporal Variation of Heavy Metals in Tree-Rings in Haatso-Atomic Road
A graph depicts the temporal change of heavy metals in tree rings at Haatso-Atomic Road. There is a general trend of increasing pollution with time. The concentrations of Fe, Zn and Cu in the tree rings are the highest in 2013, 2017 and 2018 (Fe =. 40.31 mg/kg; Cu = 9.86 mg/kg; Zn =15.78 mg/kg).
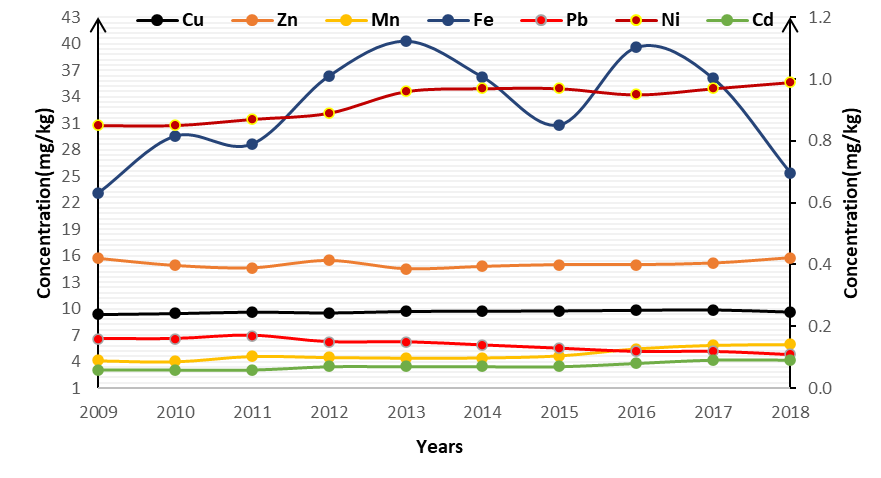
Figure2: Temporal variation of heavy metals in tree-rings at Haatso-Atomic Road
Contamination factors of heavy metals
A graph depicts the geo-accumulation index (I-geo) of heavy metals in soil at Haatso-Atomic Road. The following are the findings of the research: Cu (1.35), Mn (1.50), Pb (1.90), Zn (1.05), Ni (1.15), Cd (0.82) and Fe (3.50)
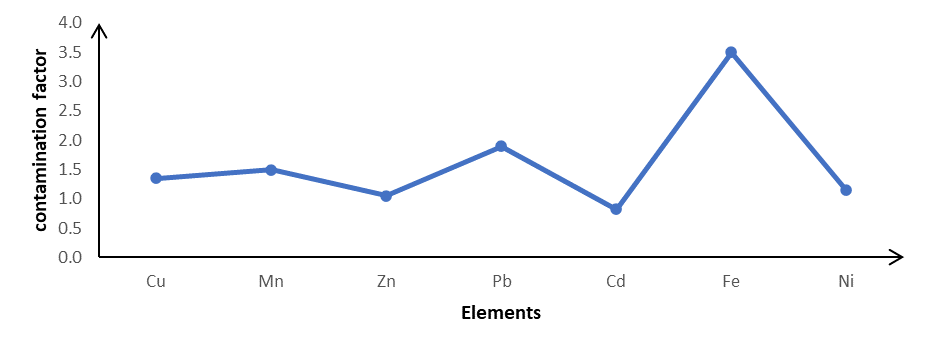
Figure3: Contamination factor of heavy metals in Haatso-Atomic Road soil.
Geo-accumulation index (I-geo) of heavy metals
At Haatso-Atomic Road, a graph shows the geo-accumulation index (I-geo) of heavy metals in soil. The study yielded the following results: Cd (-0.05), Cu (0.48), Mn (0.64), Zn (0.33), Pb (0.58), Fe (0.45), and Ni (0.53).
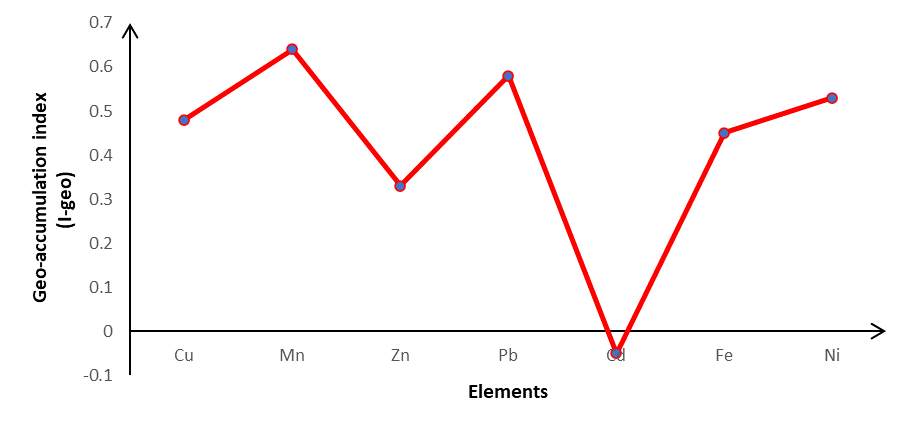
Figure4: Geo-accumulation index (I-geo) of heavy metals in soil at Haatso-Atomic Road
Enrichment Factor Analysis
At Haatso-Atomic Road, a graph displaying heavy metal enrichment factors in soil. Fe (1.0), Cd (0.0), Ni (0.0), Mn (26.2), Zn (184.8), Cu (336.7) and Pb (1797.8) are the results of the analysis
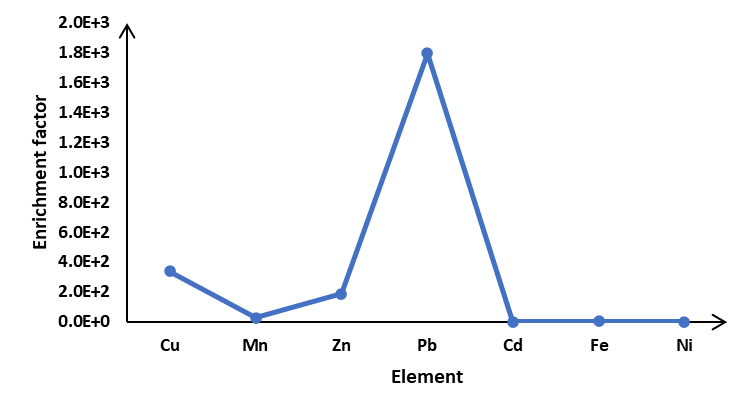
Figure5: Enrichment factors of heavy metals in soil at Haatso-Atomic Road
| Element | Ingestion | Inhalation | CR | HQ | ||||
| Child | Adult | Child | Adult | Child | Adult | Child | Adult | |
| Cu | 5.88×10−9 | 5.56×10−9 | 1.89×10−9 | 1.79×10−9 | 1.46×10−7 | 1.38×10−7 | ||
| Zn | 4.11×10−9 | 3.89×10−9 | 1.32×10−9 | 1.25×10−9 | 1.37×10−8 | 1.29×10−7 | ||
| Pb | 7.14×10−9 | 6.75×10−9 | 2.29×10−9 | 2.17×10−9 | 1.92×10−6 | 2.03×10−6 | ||
| Cd | 6.28×10−10 | 5.93×10−10 | 2.01×10−10 | 1.90×10−10 | 8.06×10−6 | 7.10×0−6 | 5.53×10−6 | 6.28×10−8 |
| Ni | 6.49×10−9 | 6.14×10−9 | 2.09×10−9 | 1.97×10−9 | 2.50×0−6 | 1.87×0−6 | 2.98×10−6 | 3.15×10−6 |
| Fe | 1.79×10−8 | 1.69×10−8 | 5.74×10−9 | 5.43×10−8 | ||||
| Mn | 7.91×10−9 | 7.84×10−9 | 2.54×10−9 | 2.40×10−9 | ||||
Table4: Health risk index of heavy metals in soils of Haatso-Atomic Road
| item | CuS | ZnS | PbS | FeS | MnS | NiS | CdS |
| CuT | 0.97 | 0.315 | 0.31 | 0.415 | 0.112 | 0.162 | 0.012 |
| ZnT | -0.023 | 0.453 | -0.067 | -0.321 | -0.421 | -0.107 | -0.41 |
| PbT | -0.301 | -0.47 | -0.241 | 0.251 | 0.041 | -0.014 | -0.051 |
| FeT | -0.621 | 0.521 | 0.762 | 0.621 | -0.21 | -0.451 | -0.11 |
| MnT | -0.514 | 0.231 | -0.621 | 0.411 | 1 | 0.015 | 0.021 |
| NiT | -0.01 | 0.031 | 0.251 | 0.31 | -0.01 | -0.141 | -0.21 |
| CdT | -0.812 | 0.071 | 0.415 | -0.033 | 0.812 | 0.712 | 0.621 |
Note: T represent in tree ring S represent in soil
Table5: Correlation between Elemental Concentrations Tree Ring and Soil at Haatso-Atomic Road
| Element | Crustal Average (ppm) | Crustal average (ug/m3) |
| Na | 23600 | 22.19 |
| Mg | 23300 | 23.16 |
| Al | 82300 | 90.82 |
| Si | 281500 | 323.36 |
| P | 1050 | 1.33 |
| S | 260 | 0.34 |
| Cl | 130 | 0.19 |
| K | 20900 | 33.42 |
| Ca | 41500 | 68.03 |
| Ti | 5700 | 11.16 |
| V | 135 | 0.28 |
| Cr | 100 | 0.21 |
| Mn | 950 | 2.1 |
| Fe | 56300 | 128.59 |
| Cu | 55 | 0.14 |
| Zn | 70 | 0.19 |
| Br | 2.5 | 0.0082 |
| Sr | 375 | 1.34 |
| Zr | 165 | 0.64 |
| Pb | 12.5 | 0.11 |
| Source: (Taylor and Mclennan, 1985) | ||
Table6: Average of crustal elements used to calculate enrichment factors of soil at Haatso-Atomic Road in the Greater Accra region in Ghana
Discussion
From Figure 2 which shows the temporal change of heavy metals in tree rings at Haatso-Atomic Road. There was a consistent increase in the levels of Fe, Zn and Cu can be attributed to increased traffic activities on the stretch of road where the samples were taken. Between different periods, the concentrations of Cd, Mn, Pb, and Ni fluctuated. Despite the observed trend, changes in heavy metal concentrations did not differ significantly between the three periods (excluding Mn), and this result was similar to that of Safo-Adu et al (2014) along the Accra-Tema route. The maximum limit of Cd, Mn, Pb and Ni in plants as recommended by WHO were higher for concentration of Cd (0.02 mg/kg) and Mn (2 mg/kg) and also lower for Pb (2.14 mg/kg) and Ni (10 mg/kg). Heavy metals in plant samples have also been measured and reported around the world. Compared with previously published reports, concentrations of Cu, Zn, Pb, and Cd (Cu, 0.93 mg/kg; Zn, 11.9 mg/kg; Pb, 0.1 5mg/kg; Cd, 0.13 mg/kg) (Odabasi et al. 2016) were lower for this study. Differences in the results may be due to different parts of plants, different species, and different environments tested. The results obtained in our study indicated that Swietenia mahagoni (mahogany) contained the highest concentrations of heavy metals and this can be attributed to traffic activities and other anthropogenic activities.
Figure 3 shows contamination factor of heavy metals in Haatso-Atomic Road soil. Using the contamination factor categories for the Haatso-Atomic roadside soil, Cu (1.35), Mn (1.50), Pb (1.90), Zn (1.05), Ni (1.15), suffered moderate contamination by all metals except Cd (0.82) which indicates a low contamination. On the other hand, Fe (3.50) had a significant amount of contamination.
Figure 4 depicts the heavy metal geo-accumulation index (I-geo) in soil along the Haatso-Atomic Road. The mean I-geo of Cd (-0.05) was less than 0, indicating that the soil was not contaminated by Cd. Cu (0.48), Mn (0.64), Zn (0.33), Pb (0.58), Fe (0.45), and Ni (0.53) all had a mean greater than0, indicating that Zn and Cd soil pollution levels were unpolluted and moderately polluted, respectively. This is due to human activities such as traffic and other anthropogenic activities.
Figure 5 shows the averages of the EFs of elements in soil at the Haatso-atomic road. At the sampling site, the elements Fe (1.0), Cd (0.0), and Ni (0.0) fall into the EF 10 category. These elements are typical of crustal origin and they were attributable predominantly to crustal or soil dust. Although these elements were mainly from natural sources, the influences of anthropogenic sources cannot be ignored. Fe is emitted from wear-and-tear of brake pads and other automobile parts (Schauer, 2006). For, Mostly, Ni has been associated with combustion of diesel which has been on the rise as the number of vehicles on the road increases (Wang et al, 2003). Cd comes from tire abrasion emissions in vehicle exhaust, open burning of municipal wastes containing Ni-Cd batteries of vehicles (Awan et al., 2011). The element Mn (26.2) belongs to the EF >10 but less than 100. Mn was moderately enriched, according to the results. Mn is also attributed to brake wear and vehicle tailpipe emission (Garg et al., 2000). Elements with EF ˃100 are Zn (184.8) and Cu (336.7) which are highly enriched with emission sources likely from traffic and refuse burning, respectively. In the group with EF ˃1000 was Pb (1797.8) and this is heavily enriched. Zn and Pb are traditional tracers of vehicle emissions and their EF values suggested a large contribution from vehicle emission. Besides fuel and motor oil combustion, brake wear, industrial emission, long-range transported dust and re-suspended soil containing deposition from previously emitted leaded gasoline could importantly sources of Pb (Yele et al., 2006). It has been reported (Miguel et al., 1997) that Zn compounds have been used extensively as antioxidants (e.g., zinc carboxylate complexes and zinc sulphonate) and as detergent or dispersant improvers for lubricating oils. Cu pollution from vehicles has been linked to wear of brake lining (Vecchi et al., 2007). Copper and zinc have been known to be good indicators of traffic emissions from brake wear and tyre emissions.
Health Risk Analysis
Heavy metals enter into the human body primarily through ingestion. HQ was less than 1, suggesting that heavy metal elements in soils of Haatso-Atomic Road pose no or little non-carcinogenic health risk to humans. Compared with adults, the HQ and CR for children were significantly higher than those for adults. Similar trends were reported by other scholars (Xiao et al. 2017). In the case of the ingestion pathway, the HQ for children was in the order of Pb>Ni>Cu>Zn>Cu>Cd; and the HQ for adults was in the order of Pb>Ni>Cd>Zn>Cu. It is reasonable to conclude that the higher ingestion rate of soil and lower body weight of children were the two reasons for the higher values of risks through ingestion (Chen et al. 2016). Encouraging proper hygienic habits can be effective in helping to reduce the ingestion rate and protect children from health risks. As shown in Table 3, the CR values for children were higher than adults. For both children and adults, the CR values followed the order of Cd>Ni. However, the CR of Cd for children and adults was 8.06 ×10−6 and 7.10 × 10−6, respectively. Thus, there was a large carcinogenic risk of Cd in Haatso-Atomic soils. The result clearly suggests that Cd was the most important risk factor and this may cause diseases such as osteoporosis and increase the risk of fractures to human beings in the study area. It is important to note that human health risk of heavy metals can vary depending on their bioavailability, which can differ with soil type and environmental factors, such as precipitation.
Conclusion
All of the heavy metal concentrations surveyed in this study were slightly higher than other reported studies. Concentrations of Fe, Zn and Cu in tree rings increased gradually in the past 10 years, and the concentration of Fe increased significantly in 2013, but the change in concentrations of Cd, Mn and Pb follow an irregular trend. Cd was found to be unpolluted, whereas Cu, Mn, Zn, Pb, Fe, and Ni were found to be unpolluted to moderate contaminated, according to I-geo's findings. Correlation analysis showed that the origin of heavy metals in soil and tree rings were associated in a difficult manner., The concentrations of Cu, Mn and Cd increase in soil and tree ring and this can be attributed to vehicular exhaust emissions and abrasion of vehicle. The main source of heavy metals (Fe, Ni, Zn and Pb) may be fertilizers, waste water and solid waste used in agriculture soil as Cd mainly originates from fertilizer and pesticides. Cd in tree rings were mainly uptake from the soil, while the other heavy metals may result from surface water uptake and air pollution. The study of sources of heavy metals in tree rings needs further research. Enrichment factor results of soil chemical components at the Haatso-Atomic Road indicate that elements of anthropogenic origin (such as Zn, Cu and Pb) were moderately and highly enriched with respect to crustal elements. The non-carcinogenic risks associated with the six heavies
metals were negligible, but the carcinogenic risk caused by Cd was significant. In terms of both non-carcinogenic and carcinogenic dangers, heavy metals put youngsters under more stress than adults. Future research should focus on the chemical behaviors of Fe in the study region's soils, which could lead to the development and implementation of appropriate remediation management strategies.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgement
I wish to acknowledge with heartfelt appreciation for all the assistance I received from the lecturers in the Physics Department of Ghana Atomic Energy Commission. I also extend my acknowledgment to the Mr. Yaw Nsuo Brobbey.
References
- Aiman, Umme, Adeel Mahmood, Sidra Waheed, and Riffat Na- seem Malik. “Enrichment, geo-accumulation and risk surveil- lance of toxic metals for different environmental compartments from Mehmood Booti dumping site, Lahore city, Pakistan.” Chemosphere 144 (2016): 2229-2237.
- Adimalla, Narsimha. “Heavy metals contamination in urban surface soils of Medak province, India, and its risk assessment and spatial distribution.” Environmental Geochemistry and Health 42, no. 1 (2020): 59-75.
- Akoto, O., J. H. Ephraim, and G. Darko. “Heavy metals pollu- tion in surface soils in the vicinity of abundant railway servicing workshop in Kumasi, Ghana.” (2009).
- Alahabadi, Ahmad, Mohammad Hassan Ehrampoush, Mo- hammad Miri, Hamideh Ebrahimi Aval, Samira Yousefzadeh, Hamid Reza Ghaffari, Ehsan Ahmadi et al. “A comparative study on capability of different tree species in accumulating heavy metals from soil and ambient air.” Chemosphere 172 (2017): 459-467.
- Awan, M. A., Ahmed, S. H., Aslam, M. R., & Qazi, I. A. (2011). Determination of total suspended particulate matter and heavy metals in ambient air of four cities in Pakistan. Journal of Ener- gy & Environment 2 (2), 128-132.
- Barbieri, Maurizio, Giuseppe Sappa, Stefania Vitale, Barbara Parisse, and Maria Battistel. “Soil control of trace metals con- centrations in landfills: A case study of the largest landfill in Eu- rope, Malagrotta, Rome.” Journal of geochemical exploration 143 (2014): 146-154.
- Barbieri, Maurizio. “The importance of enrichment factor (EF) and geoaccumulation index (Igeo) to evaluate the soil contam- ination.” Journal of Geology & Geophysics 5, no. 1 (2016): 1-4.
- Basha, Shaik, Jayaraj Jhala, Ravi Thorat, Sangita Goel, Rohit Trivedi, Kunal Shah, Gopalakrishnan Menon, Premsingh Gaur, Kalpana H. Mody, and Bhavanath Jha. “Assessment of heavy metal content in suspended particulate matter of coastal indus- trial town, Mithapur, Gujarat, India.” Atmospheric Research 97, no. 1-2 (2010): 257-265.
- Bilos, Claudio, Juan Carlos Colombo, Carlos Norberto Skorup- ka, and MJ Rodriguez Presa. “Sources, distribution and vari- ability of airborne trace metals in La Plata City area, Argentina.” Environmental pollution 111, no. 1 (2001): 149-158.
- Braga, C. F., E. C. Teixeira, L. Meira, F. Wiegand, M. L. Yonea- ma, and J. F. Dias. “Elemental composition of PM10 and PM2. 5 in urban environment in South Brazil.” Atmospheric Environ- ment 39, no. 10 (2005): 1801-1815.
- Cao, Junji, Shungcheng Lee, Xiangdong Zheng, Kinfai Ho, Xi- aoye Zhang, Hai Guo, Judith C. Chow, and Hongbin Wang. “Characterization of dust storms to Hong Kong in April 1998.” Water, Air and Soil Pollution: Focus 3 (2003): 213-229.
- Cambra, Koldo, Teresa Martínez, Arantzazu Urzelai, and Eva Alonso. “Risk analysis of a farm area near a lead-and cadmi- um-contaminated industrial site.” Journal of Soil Contamination 8, no. 5 (1999): 527-540.
- Censi, P. A. O. L. O., S. E. Spoto, F. I. L. I. P. P. O. Saiano, M. Sprovieri, Salvatore Mazzola, G. Nardone, S. I. Di Geronimo, R. Punturo, and D. Ottonello. “Heavy metals in coastal water systems. A case study from the northwestern Gulf of Thailand.” Chemosphere 64, no. 7 (2006): 1167-1176.
- Chao, C. Y., & Wong, K. K. (2002). Residential indoor PM10 and PM2. 5 in Hong Kong and the elemental composition. At- mospheric Environment, 36(2), 265-277.
- Chen, Mengqin, Lu Pi, Yan Luo, Meng Geng, Wenli Hu, Zhi Li, Shijun Su, Zhiwei Gan, and Sanglan Ding. “Grain size dis- tribution and health risk assessment of metals in outdoor dust in Chengdu, Southwestern China.” Archives of Environmental Contamination and Toxicology 70 (2016): 534-543.
- Chiarenzelli, J., L. Aspler, C. Dunn, B. Cousens, D. Ozarko, and K. Powis. “Multi-element and rare earth element composi- tion of lichens, mosses, and vascular plants from the Central Barrenlands, Nunavut, Canada.” Applied Geochemistry 16, no. 2 (2001): 245-270.
- Cocozza, C., Sonia Ravera, P. Cherubini, F. Lombardi, Marco Marchetti, and Roberto Tognetti. “Integrated biomonitoring of airborne pollutants over space and time using tree rings, bark, leaves and epiphytic lichens.” Urban forestry & urban greening 17 (2016): 177-191.
- Cui, Zhengwu, Yang Wang, Na Zhao, Rui Yu, Guanghui Xu, and Yong Yu. “Spatial distribution and risk assessment of heavy metals in paddy soils of Yongshuyu irrigation area from Songhua River Basin, Northeast China.” Chinese Geographi- cal Science 28 (2018): 797-809.
- Danek, Małgorzata, Trevor Bell, and Colin P. Laroque. “Some considerations in the reconstruction of lead levels using laser ablation: Lessons from the design stage of an urban dendro- chemistry study, St. John’s, Canada.” Geochronometria 42, no. 1 (2015): 217-231.
- Fan, L. D., M. S. Wang, D. Y. Song, and J. C. Shen. “Ecological Risk Assessment and Health Risk Assessment of Heavy Met- als in Some China Urban Parks.” Environmental Chemistry 38, no. 4 (2019): 793-804.
- Garg, Bhagwan D., Steven H. Cadle, Patricia A. Mulawa, Pe- ter J. Groblicki, Chris Laroo, and Graham A. Parr. “Brake wear particulate matter emissions.” Environmental Science & Tech- nology 34, no. 21 (2000): 4463-4469.
- Kaur, Manpreet, Ajay Kumar, Rohit Mehra, and Inderpreet Kaur. “Quantitative assessment of exposure of heavy metals in groundwater and soil on human health in Reasi district, Jam- mu and Kashmir.” Environmental geochemistry and health 42 (2020): 77-94.
- Koch, M., and W. Rotard. “On the contribution of background sources to the heavy metal content of municipal sewage sludge.” Water science and technology 43, no. 2 (2001): 67-74
- Li, Chun-Fang, Jian-Fei Cao, Jian-Shu Lü, Lei Yao, and Quan-Yuan Wu. “Ecological risk assessment of soil heavy metals for different types of land use and evaluation of human health.” Huan Jing ke Xue= Huanjing Kexue 39, no. 12 (2018): 5628-5638.
- Joshi, S. R., R. Kumar, P. Saikia, R. K. Bhagobaty, and S. Thokchom. “Impact of roadside pollution on microbial activities in subtropical forest soil of North East India.” Res J Environ Sci 4 (2010): 280-287.
- Islam, Md Saiful, Md Kawser Ahmed, Md Habibullah Al-Ma- mun, and Dennis Wayne Eaton. “Human and ecological risks of metals in soils under different land-use types in an urban environment of Bangladesh.” Pedosphere 30, no. 2 (2020): 201-213.
- de Miguel, Eduardo, Juan F. Llamas, Enrique Chacón, Torunn Berg, Steinar Larssen, Oddvar Røyset, and Marit Vadset. “Ori- gin and patterns of distribution of trace elements in street dust: unleaded petrol and urban lead.” Atmospheric Environment 31, no. 17 (1997): 2733-2740.
- Muller, G. M. M. G. M. G. M. G. P. “Index of geoaccumulation in sediments of the Rhine River.” (1969): 108-118.
- Odabasi, Mustafa, Ezgi Ozgunerge Falay, Gizem Tuna, Hasan Altiok, Melik Kara, Yetkin Dumanoglu, Abdurrahman Bayram, Doganay Tolunay, and Tolga Elbir. “Biomonitoring the spatial and historical variations of persistent organic pollutants (POPs) in an industrial region.” Environmental science & technology 49, no. 4 (2015): 2105-2114.
- Odabasi, Mustafa, Doganay Tolunay, Melik Kara, Ezgi Oz- gunerge Falay, Gizem Tuna, Hasan Altiok, Yetkin Dumanoglu, Abdurrahman Bayram, and Tolga Elbir. “Investigation of spa- tial and historical variations of air pollution around an industrial region using trace and macro elements in tree components.” Science of the Total Environment 550 (2016): 1010-1021.
- Ololade, Isaac A. “An assessment of heavy-metal contamina- tion in soils within auto-mechanic workshops using enrichment and contamination factors with geoaccumulation indexes.” Journal of Environmental protection 5, no. 11 (2014): 970-982.
- Pagotto, C., N. Remy, M. Legret, and P. Le Cloirec. “Heavy metal pollution of road dust and roadside soil near a major rural highway.” Environmental technology 22, no. 3 (2001): 307-319.
- Safo-Adu, G., F. G. Ofosu, D. Carboo, and Y. S. Armah. “Heavy metals and black carbon assessment of PM10 particulates along Accra–Tema highway in Ghana.” International Journal of Science and Technology 3, no. 8 (2014): 467-474.
- Schauer, James J., Glynis C. Lough, Martin M. Shafer, William F. Christensen, Michael F. Arndt, Jeffrey T. DeMinter, and June- Soo Park. “Characterization of metals emitted from motor ve- hicles.” Research report (Health Effects Institute) 133 (2006): 1-76.
- Speer, James H. Fundamentals of tree-ring research. Universi- ty of Arizona Press, 2010.
- Taylor, Stuart Ross, and Scott M. McLennan. “The continental crust: its composition and evolution.” (1985).
- United States Department of Agriculture (USDA); Natural Re- sources Conservation Services. Soils Quality: Urban Technical Note; (2001). Available from: http://www.nrcs.usda.gov/Inter- net/FSE_DOCUMENTS/nrcs142p2_053279.pdf
- USEPA. (1998). Risk Assessment Guidance for Superfund (RAGS). Human health evaluation manual, Interim draft. U. S. Environmental Protection Agency. USEPA. (2001). Child-Spe- cific Exposure Factors Handbook. National Center for Envi- ron-mental Assessment. EPA-600-P-00-002B.
- Vecchi, R., G. Marcazzan, and G. Valli. “A study on nighttime– daytime PM10 concentration and elemental composition in re- lation to atmospheric dispersion in the urban area of Milan (It- aly).” Atmospheric environment 41, no. 10 (2007): 2136-2144.
- Wang, Gehui, Hui Wang, Yajuan Yu, Shixiang Gao, Jianfang Feng, Songting Gao, and Liansheng Wang. “Chemical char- acterization of water-soluble components of PM10 and PM2. 5 atmospheric aerosols in five locations of Nanjing, China.” At- mospheric Environment 37, no. 21 (2003): 2893-2902.
- Watmough, Shaun A. “An evaluation of the use of dendro- chemical analyses in environmental monitoring.” Environmen- tal Reviews 5, no. 3-4 (1997): 181-201.
- Xiao, Ran, Shuang Wang, Ronghua Li, Jim J. Wang, and Ze- ngqiang Zhang. “Soil heavy metal contamination and health risks associated with artisanal gold mining in Tongguan, Shaanxi, China.” Ecotoxicology and environmental safety 141 (2017): 17-24.
- Sun, Yele, Guoshun Zhuang, Wenjie Zhang, Ying Wang, and Yahui Zhuang. “Characteristics and sources of lead pollution after phasing out leaded gasoline in Beijing.” Atmospheric En- vironment 40, no. 16 (2006): 2973-2985.
- Zhang, Yuanxun, Tauseef Quraishi, and James Jay Schauer. “Daily variations in sources of carbonaceous aerosol in Lahore, Pakistan during a high pollution spring episode.” Aerosol and Air Quality Research 8, no. 2 (2008): 130-146.


