Archive : Article / Volume 2, Issue 1
- Research Article | DOI:
- https://doi.org/10.58489/2836-5828/006
Impact of Appropriate Antibiotics within 1hr of Patients Admission
¹Grant Medical College & JJ group Of Hospitals, Mumbai, India
Sripiura Biswas, Grant Medical College & JJ group Of Hospitals, Mumbai, India
Srirupa Biswas, Sankha Dasgupta, Moumita Majumder Bhowmick, Smriti Gupta, et.al. Impact of Appropriate Antibiotics within 1hr of Patients Admission. Arch. Urol. Nephrol. (2023). Vol. 2, Iss. 1. DOI: 10.58489/2836-5828/006
© 2023 Srirupa Biswas, this is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original work is properly cited.
- Received Date: 18-02-2023
- Accepted Date: 24-02-2023
- Published Date: 01-03-2023
Antibiotics, Patients outcome, Review
Abstract
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of such infections [1,2] They may either kill or inhibit the growth of bacteria. A limited number of antibiotics also possess antiprotozoal activity [3,4]. Antibiotics are not effective against viruses such as the common cold or influenza; [5] drugs which inhibit viruses are termed antiviral drugs or antivirals rather than antibiotics.
Introduction
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of such infections.[1,2] They may either kill or inhibit the growth of bacteria. A limited number of antibiotics also possess antiprotozoal activity.[3,4] Antibiotics are not effective against viruses such as the common cold or influenza;[5] drugs which inhibit viruses are termed antiviral drugs or antivirals rather than antibiotics.
Sometimes, the term antibiotic-literally “opposing life”, from the Greek roots ἀντι anti, “against” and βίος bios, “life”-is broadly used to refer to any substance used against microbes, but in the usual medical usage, antibiotics (such as penicillin) are those produced naturally (by one microorganism fighting another), whereas non-antibiotic antibacterials (such as sulfonamides and antiseptics) are fully synthetic. However, both classes have the same goal of killing or preventing the growth of microorganisms, and both are included in antimicrobial chemotherapy. “Antibacterials” include antiseptic drugs, antibacterial soaps, and chemical disinfectants, whereas antibiotics are an important class of antibacterial used more specifically in medicine[6] and sometimes in livestock feed.
Antibiotics have been used since ancient times. Many civilizations used topical application of moldy bread, with many references to its beneficial effects arising from ancient Egypt, Nubia, China, Serbia, Greece, and Rome.[7] The first person to directly document the use of molds to treat infections was John Parkinson (1567–1650). Antibiotics revolutionized medicine in the 20th century. Alexander Fleming (1881–1955) discovered modern day penicillin in 1928, the widespread use of which proved significantly beneficial during wartime. However, the effectiveness and easy access to antibiotics have also led to their overuse[8] and some bacteria have evolved resistance to them.[1,9-11] The World Health Organization has classified antimicrobial resistance as a widespread “serious threat [that] is no longer a prediction for the future, it is happening right now in every region of the world and has the potential to affect anyone, of any age, in any country”.[12] Global deaths attributable to antimicrobial resistance numbered 1.27 million in 2019.[13]
Review Of Literature
Sepsis Campaign Guidelines 2012 and 2015 update
2015 update: “broad spectrum antibiotics should be administered within 3 hours of the time of presentation”
2012 Guideline: “We recommend that empiric antimicrobials be administered within 1 hr of the identification of severe sepsis. Blood cultures should be obtained before administering antibiotics when possible, but this should not delay initiation of antibiotics. The empiric drug choice should be changed as epidemic and endemic ecologies dictate (eg, H1N1, methicillin-resistant S. aureus, chloroquine-resistant malaria, penicillin-resistant pneumococci, recent ICU stay, neutropenia) (grade 1D)”
As of 2010, only 68% of patients in the SSC registry received antibiotics within 3 hours.
Kumar et al, 2006
The classic retrospective cohort study of 2,731 septic shock ICU patients showed a strong correlation between delay in effective antibiotic therapy and in-hospital mortality after recurrent or persistent arterial hypotension (P <0.0001) decrease in survival by 7.6% for the delay of 1 hour of antibiotic therapy over the ensuing 6 hours.
Only 50% of the patients received effective antibiotic therapy within the first 6 hours (i.e. appropriate in vitro activity for the isolated pathogenic microorganism or the underlying clinical syndrome).
Gaieski et al, 2010
A single-center cohort study of 261 patients with severe sepsis undergoing early goal-directed therapy (EGDT) time from triage and qualification for EGDT to appropriate antibiotic therapy was significantly associated with reduced mortality at the <1 hour cutoff point (odds ratio = 0.3 and 0.5, each P <0.03)
Puskarich et al, 2011
A preplanned analysis of a multicenter controlled trial (3 centers) in US EDs of 291 patients with septic shock (EMSHOCK NET) found no change in mortality with hourly delayed antibiotic therapy up to 6 hours after triage or after recognition of shock.
Antibiotic administration before recognition of shock was associated with a lower mortality as compared with antibiotic administration after recognition of shock (odds ratio = 2.35, 95%CI 1.12 to 4.53).
Ferrer et al, 2014
Retrospective analysis of 17,990 patients with severe sepsis and septic shock from the multicenter, multinational Surviving Sepsis Campaign database (Europe, USA and South America) found an hourly increase in mortality with delay in antibiotic administration following recognition of severe sepsis, not just the onset of hypotension
Differences were statistically significant, as we as clinically significant, beyond 2 hours
De Groot et al, 2015
Prospective multicenter study in three Dutch Eds,
1,168 patients with sepsis (stratified into mild, moderate and severe; overall mortality of 10%)
in those receiving antibiotics within 6 hours, a reduction in time to antibiotics was not found to be associated with an improvement in relevant clinical outcomes (28 mortality or LOS)
Sterling et al, 2015
Systematic review of 11 studies that met inclusion criteria, comprising:
16,178 patients with severe sepsis/ septic shock who evaluable for antibiotic administration from emergency department triage, and 11,017 patients who were evaluable for antibiotic administration from severe sepsis/septic shock recognition
There was no significant mortality benefit of administering antibiotics within 3 hours of ED triage or within 1 hour of shock recognition in severe sepsis and septic shock
The authors suggest that currently recommended timing metrics as measures of quality of care are not supported by the available evidence
Problems: 7 studies were excluded because authors did not respond to requests for information (selection bias), unclear if antibiotic choice was appropriate in these studies and less than half of patients had confirmed bacteraemia (antibiotics would not be expected to benefit non-bacterial infections or infections with bacteria that are insensitive to the chosen antibiotic)
Garnacho-Montero et al, 2015
A prospective obsertional study of 928 patients admitted to ICU with severe sepsis/ septic shock (68% with microbiological identification)
Findings were:
Inadequate therapy prior to ICU admission was more common in nosocomial sepsis
Administration of appropriate empirical antimicrobial therapy early was associated with decreased mortality
Nearly all patients (98.3 %) received at least one dose of antibiotics before ICU admission, however they were inadequate in 31% of patients
Progression to septic shock in patients with severe sepsis was associated with inadequate antimicrobial therapy prior to ICU admission
Siriwimon Tantarattanapong et al,2021
The current international sepsis guideline recommends that administration of intravenous broad-spectrum antibiotics should be initiated within 1 hour of emergency department (ED) arrival for sepsis patients.
Retrospective cross-sectional study, elderly patients (age ¸65 years) diagnosed with sepsis in the ED of a tertiary referral and academic hospital from January were enrolled. Door-to-antibiotic time was defined as the time from ED arrival to antibiotic initiation. The associations of door-to-antibiotic time and each hour delay in first antibiotic initiation with in-hospital mortality were assessed.
Six hundred patients with the median age of 78.0 (IQR: 72.0-86.0) were studied (50.8% female). The median door-to-antibiotic time was 51.0 (36.0–89.0) minutes and in-hospital mortality rate was 12.5%. There was no significant difference in the in-hospital mortality rate between door-to-antibiotic time ·1hour and >1 hour (13.1% vs. 11.6%, p = 0.726). When considering hour-upon-hour of door-to-antibiotic time, no significant difference in in-hospital mortality was observed (p = 0.866). Factors that led to a delay in door to-antibiotic time were presenting body temperature <38°C (odds ratio [OR] 3.34; 95% CI, 2.12-5.29; p < 0.001) and age <75 years (OR 1.7; 95% CI, 1.09-2.64; p= 0.019).
Door-to-antibiotic time was not associated with in-hospital mortality in elderly sepsis patients in this study. Significant factors that led to a delay in
door-to-antibiotic time were no fever, age <75 years, doctor time, and blood sample taking time.
Michael G. Allison et al,2016
Although early retrospective studies found decreased survival associated with each 1-hour delay in antibiotics, prospective studies have not validated these findings; the optimal time benefit of antibiotic delivery within the first 6 hours is not known. Inappropriate initial antibiotic therapy is associated with an increase in mortality; it is appropriate to start broad-spectrum antibiotic therapy that provides coverage of the most likely pathogens.
The use of 2 antibiotics to double-cover gram-negative infections is not routinely required, especially if empiric therapy involves an antipseudomonal penicillin, cephalosporin, or carbapenems.
Patients who receive both vancomycin and piperacillin/tazobactam may be at greater risk for acute kidney injury. The loading dose of antibiotics is the same in patients with and without renal dysfunction. Subsequent doses need to be adjusted in patients with renal dysfunction.
Peter M. Houck, et al,2014
Pneumonia accounts for more than 600000 Medicare hospitalizations yearly. Guidelines have recommended antibiotic treatment within 8 hours of arrival at the hospital.
Retrospective study using medical records from a national random sample of 18209 Medicare patients older than 65 years who were hospitalized with community-acquired pneumonia from July 998 through March 1999. Outcomes were severity adjusted mortality, readmission within 30 days of discharge, and length of stay (LOS).
Among 13771 (75.6%) patients who had not received outpatient antibiotic agents, antibiotic administration within 4 hours of arrival at the hospital was associated with reduced in-hospital mortality (6.8% vs 7.4%; adjusted odds ratio [AOR], 0.85; 95% confidence interval [CI], 0.74-0.98), mortality within 30 days of admission (11.6% vs 12.7%; AOR, 0.85; 95% CI, 0.76-0.95), and LOS exceeding the 5-day median (42.1% vs 45.1%; AOR, 0.90; 95% CI, 0.83-0.96). Mean LOS was 0.4 days shorter with antibiotic administration within 4 hours than with later administration. Timing was not associated with readmission. Antibiotic administration within 4 hours of arrival was documented for 60.9% of all patients and for more than 50% of patients regardless of hospital characteristics.
Antibiotic administration within 4 hours of arrival was associated with decreased mortality and LOS among a random sample of older inpatients with community-acquired pneumonia who had not received antibiotics as outpatients. Administration within 4 hours can prevent deaths in the Medicare population, offers cost savings for hospitals, and is feasible for most inpatients.
Terrence Endres et al,2018
Well established in the Emergency Department (ED) literature is that the most important factor in decreasing subsequent infection rate in open fractures is the time to first administration of antibiotics. As such, the authors developed a new ED open fracture antibiotic protocol to facilitate more expeditious antibiotic administration and appropriate choice of antibiotics.
During Phase 1 of this project, the authors identified the 2012 - 2016 historical length of time from presentation of an open fracture to the possible initiation of antibiotic therapy at their institution. Results demonstrated critical areas for improvement in both timing and types of antibiotics administered. Phase 2 of the study evaluated the effect of the new open fracture antibiotic protocol. Sample cases from both phases were then further identified based on type of open fracture, time to initiation of antibiotics from ED presentation, type of antibiotics, and time to definitive treatment. Analyses were performed using GraphPad proprietary software.
A random sample of 110 patients were included from Phase 1 and 27 patients from Phase 2. A total of 43 Phase 1 patients were administered cefazolin (Kefzol, Ancef); the remainder of the patients received a number of different antibiotics. During Phase 2, all 27 patients received cefazolin and Gentamycin if necessary per the new protocol. The average time to initiation of antibiotics was 0.907 hours during Phase 1 compared to 0.568 hours in Phase 2. The new protocol also significantly decreased the average time to antibiotics in ED from 2.17 hours to 1.82 hours when including EMS transfer time. Average time to definitive treatment in the operating room was 6.63 hours during Phase 1 and was significantly lowered to 3.97 hours during Phase 2.
Timing to initiation of antibiotics after open fractures is the most important aspect to decrease infection rates. In order to decrease these times, the authors implemented a new ED protocol that specifically stated the type of antibiotic to be given based on the open fracture without orthopedics needing to be notified before administration. Ideally, the use of such protocols in ED settings will serve to greatly decrease infection risks after open fracture.
Gabor Zilahi et al,2016
There are certain well defined clinical situations where prolonged therapy is beneficial, but prolonged duration of antibiotic therapy is associated with increased resistance, medicalizing effects, high costs and adverse drug reactions.
The best way to decrease antibiotic duration is both to stop antibiotics when not needed (sterile invasive cultures with clinical improvement), not to start antibiotics when not indicated (treating colonization) and keep the antibiotic course as short as possible. The optimal duration of antimicrobial treatment for ventilator-associated pneumonia (VAP) is unknown, however, there is growing evidence that reduction in the length of antibiotic courses to 7–8 days can minimize the consequences of antibiotic overuse in critical care, including antibiotic resistance, adverse effects, collateral damage and costs.
Biomarkers like C-reactive protein (CRP) and procalcitonin (PCT) do have a valuable role in helping guide antibiotic duration but should be interpreted cautiously in the context of the clinical situation. On the other hand, microbiological criteria alone are not reliable and should not be used to justify a prolonged antibiotic course, as clinical cure does not equate to microbiological eradication.
We do not recommend a ‘one size fits all’ approach and in some clinical situations, including infection with non-fermenting Gram-negative bacilli (NF-GNB) clinical evaluation is needed but shortening the antibiotic course is an effective and safe way to decrease inappropriate antibiotic exposure.
María Luisa Martínez et al, 2020
Sepsis is a medical emergency and life-threatening condition due to a dysregulated host response to infection, which is time-dependent and associated with unacceptably high mortality. Thus, when treating suspicious or confirmed cases of sepsis, clinicians must initiate broad-spectrum antimicrobials within the first hour of diagnosis.
Optimizing antibiotic use is essential to ensure successful outcomes and to reduce adverse antibiotic effects, as well as preventing drug resistance. All likely pathogens involved should be considered to provide an appropriate antibiotic coverage. Clinicians must investigate on the previous risk of multidrug-resistant (MDR) pathogens, and the principle of individualized dosing should replace the principle of standard dosing.
The loading dose is an initial higher dose of an antibiotic for all patients, yet an individualized treatment approach for further doses should be implemented according to pharmacokinetics (PK)/pharmacodynamics (PD) and the presence of renal/liver dysfunction. Extended or continuous infusion of beta-lactams and therapeutic drug monitoring (TDM) can help to achieve therapeutic levels of antimicrobials. Reevaluation of duration and appropriateness of treatment at regular intervals are also necessary. De-escalation and shortened courses of antimicrobials must be considered for most patients, except in some justified circumstances.
Leadership, teamwork, antimicrobial stewardship (AS) frameworks, guideline’s recommendations on the optimal duration of treatments, de-escalation, and novel diagnostic stewardship approaches will help us to improve patients’ quality of care.
Balasubramanian Venkatesh et al, 2021
Sepsis bundles, promulgated by Surviving Sepsis Campaign have not been widely adopted because of variability in sepsis identification strategies, implementation challenges, concerns about excess antimicrobial use, and limited evidence of benefit.
1-hour septic shock and a 3-hour sepsis bundle were implemented using a Breakthrough Series Collaborative in 14 public hospitals in Queensland, Australia. A before (baseline) and after (post-intervention) study evaluated its impact on outcomes and antimicrobial prescription in patients with confirmed bacteremia and sepsis. Findings Between 01 July 2017 to 31 March 2020, of 6976 adults presenting to the Emergency Departments and had a blood culture taken, 1802 patients (732 baseline, 1070 post-intervention) met inclusion criteria. Time to antibiotics in 1- hour 73.7% vs 85.1% (OR 1.9 [95%CI 1.1-3.6]) and the 3-hour bundle compliance (48.2% to 63.3%, OR 1.7, [95%CI 1.4 to 2.1]) improved post-intervention, accompanied by a significant reduction in Intensive Care Unit (ICU) admission rates (26.5% vs 17.5% (OR 0.5, [95%CI 0.4 to 0.7]). There were no significant differences in-hospital and 30-day post discharge mortality between the two phases. In a post-hoc analysis of the post-intervention phase, sepsis pathway compliance was associated with lower in-hospital mortality (9.7% vs 14.9%, OR 0.6, 95%CI 0.4 to 0.8). The proportions of appropriate antimicrobial prescription at baseline and post-intervention respectively were 55.4% vs 64.1%, (OR 1.4 [95%CI 0.9 to 2.1]).
1-hour and 3-hour sepsis bundles for patients presenting with bacteremia resulted in improved bundle compliance and a reduced need for ICU admission without adversely influencing antimicrobial prescription.
Sandra A. Asner et al, 2021
The impact of the timeliness of antibiotic therapy on the outcome of patients with sepsis or septic shock. Methods: We searched MEDLINE, EMBASE, the Cochrane Library, Web of Science, Open-SIGLE databases, ClinicalTrials.gov and the meta-Register of Controlled Trials on July 27, 2020 for relevant studies on the timing of antibiotic therapy in adult patients with sepsis or septic shock. The primary outcome measure was all-cause crude or adjusted mortality at reported time points.
Included 35 sepsis studies involving 154,330 patients. Nineteen studies (54%) provided in- formation on the appropriateness of antibiotic therapy in 20,062 patients of whom 16,652 patients (83%) received appropriate antibiotics. Twenty-four studies (68.6%) reported an association between time-to- antibiotics and mortality. Time thresholds associated with patient’s outcome varied considerably between studies consisting of a wide range of time cutoffs (1 h, 125 min, 3 h or 6 h) in 14 studies, hourly delays (derived from the analyses of time intervals ranging from to 1 to 24 h) in 8 studies or time-to-antibiotic in 2 studies. Analyses of subsets of studies that focused on patients with septic shock (11 studies, 12,756 patients) or with sepsis (6 studies, 24,281 patients) yielded similar results.
While two-thirds of sepsis studies reported an association between early administration of antibiotic therapy and patient outcome, the time-to-antibitiocs metrics varied significantly across studies and no robust time thresholds emerged.
Toshikazu Abe et al,2019
Time to antibiotic administration is a key element in sepsis care; however, it is difficult to implement sepsis care bundles. Additionally, sepsis is different from other emergent conditions including acute coronary syndrome, stroke, or trauma. We aimed to describe the association between time to antibiotic administration and outcomes in patients with severe sepsis and septic shock in Japan.
Prospective observational study enrolled 1184 adult patients diagnosed with severe sepsis based on the Sepsis-2 criteria and admitted to 59 intensive care units (ICUs) in Japan between January 1, 2016, and March 31, 2017, as the sepsis cohort of the Focused Outcomes Research in Emergency Care in Acute Respiratory Distress Syndrome, Sepsis and Trauma (FORECAST) study.
Compared the characteristics and in-hospital mortality of patients administered with antibiotics at varying durations after sepsis recognition, i.e., 0–60, 61–120, 121–180, 181–240, 241–360, and 361–1440 min, and estimated the impact of antibiotic timing on risk-adjusted in-hospital mortality using the generalized estimating equation model (GEE) with an exchangeable, within-group correlation matrix, with “hospital” as the grouping variable.
Marin H. Kollef et al,2021
Severe or life-threatening infections are common among patients in the intensive care unit (ICU). Most infections in the ICU are bacterial or fungal in origin and require antimicrobial therapy for clinical resolution. Antibiotics are the cornerstone of therapy for infected critically ill patients. However, antibiotics are often not optimally administered
resulting in less favorable patient outcomes including greater mortality. The timing of antibiotics in patients with life threatening infections including sepsis and septic shock is now recognized as one of the most important determinants of survival for this population. Individuals who have a delay in the administration of antibiotic therapy for serious infections can have a doubling or more in their mortality. Additionally, the timing of an appropriate antibiotic regimen, one that is active against the offending pathogens based on in vitro susceptibility, also influences survival.
Not only is early empiric antibiotic administration important but the selection of those agents is crucial as well. The duration of antibiotic infusions, especially for β-lactams, can also influence antibiotic efficacy by increasing
antimicrobial drug exposure for the offending pathogen. However, due to mounting antibiotic resistance, aggressive antimicrobial de-escalation based on microbiology results is necessary to counterbalance the pressures of early
broad-spectrum antibiotic therapy. In this review, we examine time related variables impacting antibiotic optimization as it relates to the treatment of life-threatening infections in the ICU.
In addition to highlighting the importance of antibiotic timing in the ICU we hope to provide an approach to antimicrobials that also minimizes the unnecessary use of these agents. Such approaches will increasingly be linked to advances in molecular microbiology testing and artificial intelligence/machine learning. Such advances should help identify patients needing empiric antibiotic therapy at an earlier time point as well as the specific antibiotics required in order to avoid unnecessary administration of broad-spectrum antibiotics.
Vincent X. Liu et al.2017
Association between antibiotic timing and mortality among patients with sepsis receiving antibiotics within 6 hours of emergency department registration.
Retrospective study of 35,000 randomly selected inpatients with sepsis treated at 21 emergency departments between 2010 and 2013 in Northern California. The primary exposure was antibiotics given within 6 hours of emergency department registration. The primary outcome was adjusted in-hospital mortality. We used detailed physiologic data to quantify severity of illness within 1 hour of registration and logistic regression to estimate the odds of hospital mortality based on antibiotic timing and patient factors.
The median time to antibiotic administration was 2.1 hours (interquartile range, 1.4–3.1 h). The adjusted odds ratio for hospital mortality based on each hour of delay in antibiotics after registration was 1.09 (95% confidence interval [CI], 1.05–1.13) for each elapsed hour between registration and antibiotic administration. The increase in absolute mortality associated with an hour’s delay in antibiotic administration was 0.3% (95% CI, 0.01–0.6%; P = 0.04) for sepsis, 0.4% (95% CI, 0.1–0.8%; P = 0.02) for severe sepsis, and 1.8% (95% CI, 0.8–3.0%; P = 0.001) for shock.
In a large, contemporary, and multicenter sample of patients with sepsis in the emergency department, hourly delays in antibiotic administration were associated with increased odds of hospital mortality even among patients who received antibiotics within 6 hours. The odds increased within each sepsis severity strata, and the increased odds of mortality were greatest in septic shock.
Marin H Kollef, 2021, Washington
Retrospective multicentre cohort study with 15183 adults patients having sepsis & positive clinical cultures.
Delay use of appropriate antibiotics (>72 hrs) increase the fungal & bacterial infections as well as mortality 10%.
PK/PD targets were highly variable among the different antibiotics ranging from35% for an aggressive target and 78.9% for traditionally acceptable target.
Microbiology results are necessary to counterbalance the pressure of early broad-spectrum antibiotic therapy.
ICU is a particularly attractive target for AI/ML based analysis but cannot replace the role of rapid molecular testing.
Among 15183 cases, 12398(81.6%) received appropriate antibiotics.
Among 1995 patients with community acquired pneumonia of which 39.7% received broad spectrum antibiotics but recovered only 3%.
H. SEOK, 2019, KOREA
The prospective cohort study with 482 diagnosed sepsis & high grade infection patients.
COX proportional hazards regression analysis was used.
The time to antibiotics administration did not significantly affect the 7-, 14-, &28- day’s mortality in the multivariate analysis.
The study method is as like as our study.
The mortality rate:
52% -received antibiotics within 3 hours (74 patients)
43% - received antibiotics after 3 hours (340patients)
49.3% - received antibiotics within 6 hours (450 patients)
50%- received antibiotics after 6 hours. (450 patients)
YOUNG MIN JOO, 2014, SEOL, KOREA
This is a prospective cohort study.
IQR (Inter Quartile Range) & the WILCOXAN rank sum test were used for comparison.
Method is just like our study
Multivariate Linear Regression analysis showed that timely antibiotic administration was significantly associated with increased delta SOFA & decreased hospital LOS.
Among 591 patients 63.8% received timely antibiotic, 36.2% patients received delayed antibiotic.
TOSHIKAZU ABE, 2019, JAPAN
This is a prospective study enrolled 1184 adults’ patients diagnosed with severe sepsis based on the sepsis-2 criteria & admitted to ICU.
This study shows the only randomized controlled trial (RCT) that evaluated early antibiotic use in patients with suspected infection failed to reduce mortality.
FORECAST (Focused outcomes Research in emergency care in acute Respiratory Distress, syndrome, sepsis & Trauma) database is used.
The primary outcome was in hospital mortality.
The secondary outcomes were VFD & ICU free days.
Three phases in the clinical course of sepsis are recognized:
Time from onset of infection to a detectable to diagnosis.
Time from a detectable condition to diagnosis.
Time from diagnosis to antibiotic administration
Among 1124 patients 30.5% received antibiotic within 1 hour. 73.9% of the patients received antibiotic within 3 hours.
JAHANA ASCUNTAR, 2020, COLOMBIA
(Antimicrobial administration time in patients with suspected sepsis).
This is a multicenter prospective cohort study.
A standardized mean difference (SMD) <10% was considered an adequate balance in the distribution between the group.
Method is just like our method.
The sepsis-3 criteria were not part of the study population collection process.
The study based on total eligible population with any organ dysfunction.
Primary purpose: estimate the effect of each component of EGDT protocol.
Bethany A Kalich et all2012
Determine impact of antibiotic specific sepsis bundle on timely initiation of appropriate antibiotics.
Prospective cohort study of patients of tertiary care hospital admitted with severe sepsis
It was done from October-december,2012.
Total patients-1023(randomly selected and only sepsis patients)
Appropriate antibiotic was 33.9% vs 54.8%, within 1h in 22.6% vs 14.5%
All appropriate antibiotics were initiated in 16.1% vs 12.9% of patients within 3h in 14,5% vs 9.7%
Conclusion- Improvement in results of appropriate initial antibiotic therapy for severe sepsis.
Our study is different as we take all patients who got antibiotic within 0-24h of admission in ICU.
Garnacho-montero et all2009
Evaluate impact of antibiotic and mortility rate of patients admitted in ICU with sepsis
Prospective cohort study done in ICU of tertiary hospital.
All patients taken who were suffering from sepsis.
Total patient-406, sepsis-105(25.9%), severe sepsis-116(28.6%), septic shock-185(45.6%)
SOFA score at ICU admission (OR-1.29,95%, CONFIDENCE INTERVAL-1.19-1.40)
Inadequate anti-microbial therapy 8.14,95%, CI- 1.98-33.5
Adequate anti-microbial therapy (OR-0.37,95%, CI-0.18-0.77) UROSEPSIS (OR-0.37,95%, CI-0.05-0.41)
CONCLUSION- In patients admitted in ICU for sepsis, the adequacy of empirical anti-microbial treatment is crucial in terms of outcome.
Sandra.A.Asner et all ,2021
Review the impact of timeliness of antibiotic therapy on outcome of patients with sepsis and septic shock
Search based study from Search Medline, Embase, Web of science, Meta Register of controlled trials on July 27,2020
Include 35 sepsis studies involving 154,336 patients.
19 studies provided information on appropriateness of antibiotic therapy in 20,062 patients of whom 16,652 patients receive appropriate antibiotics.
24 studies report associate between time to time antibiotics and mortality.
Time interval taken from 1-24 h.
Conclusion- Timely and appropriate antibiotic is essential for speedy recovery and reducing mortality.
Michael A Puskarich et all2011
Multicenter randomized controlled study to determine association between time to initial antibiotics and mortality of septic shock patient.
3 Urban US ED was taken.
Central venous pressure, Mean arterial pressure and central venous oxygen saturation are the interventions.
291 patients were included
1 hour (OR-1.2,0.6-2.5), 2hours (OR-0.71,0.4-1.3), 3hours (OR-0.59,0.3-1.3)
Mortality was increased patients received initial antibiotic after shock recognition(N=172,59%) compared with before shock (OR-2.4,1.1-4.5)
No increase mortality with each hour delay to administration of antibiotics after triage. Delay in antibiotics until after shock recognition was associated with increased mortality.
Shahla Siddiqui et all2000
To access the promptness of antibiotic administration to patients with sepsis and effects on survival.
4 months prospective study at ED of Aga Khan University hospital.
111 patients were included. Among them severe sepsis-52%,most common organism was found –Salmonella typhi
100 patients received intravenous antibiotics at ED.(F statistics 2.17,p=0.003)
Using COX regression model determined that survival dropped acutely with every hourly delay in antibiotic administration.
Each hourly delay in antibiotic administration was associated with an increase in mortality.
AIMS & OBJECTIVE
- The mantra for timing of antibiotics for serious infections is ‘hit hard, early and appropriately’
- Evaluate outcome of patients admitted in hospital how receive an appropriate antibiotic within 1hour of admission.
- Despite the strong biological plausibility of a need for early antibiotics in patients with serious bacterial infections the importance of antibiotic timing is controversial.
Rationale for Early Antibiotics
- Early antibiotics may:
- prevent injury caused by microbial activity and toxin production
- prevent or ameliorate harmful host responses to infection
- Observational data has shown strong associations between early antibiotics and survival outcomes; however, a recent (flawed) systematic review did not find a benefit for early antibiotics.
- The Sequential Organ Failure Assessment (SOFA) score numerically quantifies the number and severity of failed organs. We examined the utility of the SOFA score for assessing outcome of patients with antibiotics at the time of intensive care unit (ICU) presentation.
- APACHE IV provided the best discrimination and calibration abilities and was useful for quality assessment and predicting mortality in medical ICU patients with antibiotics.
Methods

.
STUDY LOCATION: AMRI HOSPITAL -DHAKURIA,
- Timing of ICU admission - time noted from the patient’s ICU nursing chart when the first vital parameters are noted by the nurse.
- Timing of administration of 1st dose of antibiotic will be noted from ICU nursing chart (in minutes from the timing of ICU admission).
- Antibiotic that is administered within the first one hour of admission will be noted from ICU nursing chart.
- Appropriateness of antibiotic - will be assessed from the microbiology culture and sensitivity results once available.
- Outcome measures - will be noted from the ICU database.
- Data will be then recorded systemically in the data collection form and finally entered in the excel sheet for analysis.
- All the data will be then analysed by statistician by appropriate statistical tests.
Search strategy:
The data search was performed on July 22, 2022 till November 28, 2022 in AMRI HOSPITAL -DHAKURIA, we conducted a prospective study of adult patients in ICU ward. Records screened “Patient who are present in ICU for more than 24 hours with antibiotics”. The ICU ward which was included in the study was ICU-2 with 11 beds, ICU -3 with 12 beds, ICU -7 with 9 beds and NS-ICU with 8 beds. Ethical approval was obtained from the institutional review board. Demographic, clinical, and study data were recorded from charts through the electronic medical.
Data extraction and eligibility criteria:
Patients were excluded if they met any of the following criteria
- <18>
- pregnant or post-delivery,
- Excluded If Not Prescribed Antibiotic.
- Excluded If Antibiotic Were Prescribed More Than 24h After Time Zero.
- Discharge against medical advice (DAMA) Excluded
- TSL (Temperament suspension of life support) excluded.
- If a pathogen sample is not sent for none of the culture test like (blood culture, urine culture or sputum culture).
Patient repots inclusion criteria: -
- Patients ≥ 18 years old.
- If a pathogen sample is sent for any of the culture test like (blood culture, urine culture or sputum culture).
Patient characteristics:
- Patients received antibiotic < 1>
- Patients received antibiotic > 1 hour, P value
We recorded information on the selection of patients, inclusion criteria, the duration and time period of the study, the setting (intensive care units), the study design and the total number of patients received antibiotics. We also extracted data on other key study characteristics such as the set point intervals used for assessing the timing and impact of antibiotic therapy, the assessment of the appropriateness of antibiotic therapy and the study endpoints. Criteria used for the analysis of antibiotic appropriateness were based on in vitro susceptibility of causative pathogens in case of microbiologically-documented infections or on antibiotic therapy management guidelines in case of clinically-documented infections.
Outcome measures:
The primary outcome was all-cause mortality and length of stay (ICU & hospital) at the time points reported in the study. Such as INP number, gender, age, admission status (time) cause of admission, date of admission, name of antibiotic ,use of antibiotic number hour of admission, Time of antibiotic received, ITU stay, ITU outcome, Hospital outcome, Total stay, use of mechanical ventilation, Number of day on mechanical ventilation, APACHE IV, SOFA, Co-morbidities (Hypertension, II diabetes, Renal disease, Malignancy, Lung disease, Liver disease, Cancer, Heart disease, Other comorbidities),blood culture, urine culture, sputum culture, appropriate antibiotics , mortality.
Statistical Analysis
The outcome of this study was the association of clinical outcomes of infection with early antibiotics use. For the purposes of this study, early antibiotics use was defined as the time interval from ICU triage to the administration of broad-spectrum antibiotics within one hour. Those who received antibiotic administration more than one hour from ICU triage will be classified as having late antibiotics use. Broad-spectrum antibiotics referred to antibiotics that were effective against both gram-positive and gram-negative bacteria. In the included data, broad-spectrum antibiotics referred to b-lactamase-inhibit penicillin combined with third-generation cephalosporins. We applied the T-test to hospital length of stay and used chi-square to determine the correlation of early antibiotics use to mortality, mechanical ventilation support, and ICU admission. ICU admission was defined as direct admission to the ICU from the ED. We also performed factor analysis regarding the timing of antibiotics administration, including patients’ gender, age, vital signs at ICU triage, clinical symptoms.
The final sample size was 145 patients in the study. Continuous data are demonstrated as median with mean ± standard deviation. Categorical data are presented as number and percentages. The Pearson’s chi-squared test was performed on categorical data for the primary outcome. The chi-square test was used for the analysis and to compare mortality and antibiotic received time interval at ≤1 hour and received beyond the first hour. A two-sided p- value <0>
Ethical approval
- Ethical approval was taken from the AMRI Ethics Committee prior to data collection process.
- Apps used for sofa calculation is MDCalc
Result
Characteristics of the study population:
There was a total of 145 patients (n=145) admitted during study period. 63, 43.4% patients were admitted receive antibiotic were female and the rest 82, 56.6% male received antibiotic. Male patients received antibiotic within one hour 26, 52.0 % and male patients received more than one hour 56, 58.9%. Female patients received antibiotic within one hour is 24, 48.0% and Female patients received antibiotic more than one hour is 39,41.1%. (Figure 1). There was no significant difference in the gender of the patients as the p value is > 0.05 the P value is 0.422 The patients age group was divided into 7 groups 31-40 (patients received antibiotic within one hour 0,0.0% and patients received more than one hour 5,5.3%) ,41-50(patients received antibiotic within one hour 2,4.0% and patients received more than one hour 6,6.3%) 51-60(patients received antibiotic within one hour 10,20.0% and patients received more than one hour 17,17.9%),61-70 (patients received antibiotic within one hour 13,26.0% and patients received more than one hour 33, 34.7%),71-80 (patients received antibiotic within one hour 17, 34.0% and patients received more than one hour 24,25.3%),81-90 (patients received antibiotic within one hour 8,16.0% and patients received more than one hour 9,9.5%),91-100 (patients received antibiotic within one hour 0,0.0% and patients received more than one hour 1,1.1%). There was no significant difference in the age of the patients as the p value is > 0.05 the P value is 0.378. The mean APACHE IV score of patients received antibiotic within one hour 77.70 and patients received more than one hour 76.28 and the mean SOFA patients received antibiotic within one hour 3.52 and patients received antibiotic more than one hour 3.53. There was no significant difference in the APACHE IV score of the patients as the p value is > 0.05 the P value is 0.469. There was no significant difference in the SOFA score of the patients as the p value is > 0.05 the P value is 0.846. The baseline characteristics of the study population are described in Table 1.
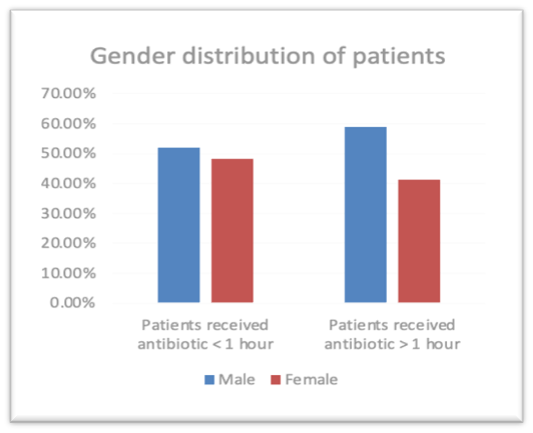
Figure 1: Gender distribution of patients who have received Antibiotic.
| GENDER | Frequency | Percent |
| FEMALE | 63 | 43.4 |
| MALE | 82 | 56.6 |
| Total | 145 | 100.0 |
| AGE | Frequency | Percent |
| 31-40 | 5 | 3.4 |
| 41-50 | 8 | 5.5 |
| 51-60 | 27 | 18.6 |
| 61-70 | 46 | 31.7 |
| 71-80 | 41 | 28.3 |
| 81-90 | 17 | 11.7 |
| 91-100 | 1 | .7 |
| Total | 145 | 100.0 |
| Characteristics | Patients received antibiotic < 1> | Patients received antibiotic > 1 hour | P value |
| Age, mean ± SD | 69.66±11.68 | 66.06±12.50 | 0.115 |
| Gender, n (%) | |||
| Male | 52.0% | 58.9% | 0.422 |
| Female | 48.0% | 41.1% | |
| APACHE IV score, mean ± SD | 77.70±19.71 | 76.28±16.05 | 0.469 |
| OFA score, mean ± SD | 3.52±0.93 | 3.53±0.83 | 0.846 |
Table 1: Showing the baseline characteristics of the study population.
Co-Morbidities
The Co-morbidities are another variable which impact the study frequency and percentage are calculation also hypertension is a common co-morbidities 115 ,79.3%, diabetes 71,49%, renal disease 3,2.1% lung disease 6,4.1% heart disease 9,6.2%. (Table 2) There was no significant difference in the co-morbidities of the patients as the p value is > 0.05 but in case of hypertension the p value is > 0.05, P value is 0.045. (Figure 2)
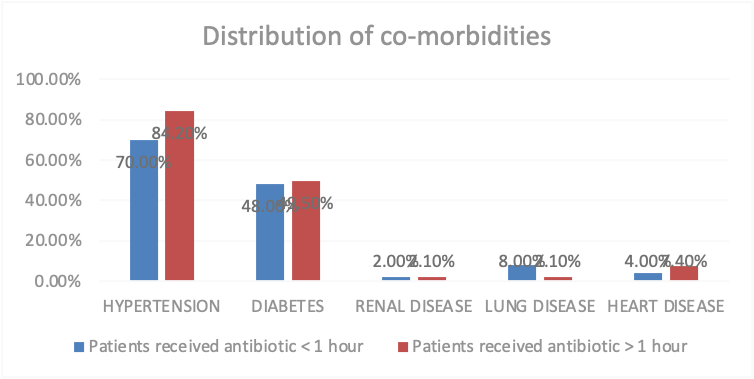
Figure 2: Distribution of co-morbidities of patients who have received Antibiotic
|
CO-MORBIDITICS DIABETES |
Frequency |
Percent |
|
NO |
74 |
51.0 |
|
YES |
71 |
49.0 |
|
Total |
145 |
100.0 |
|
CO-MORBIDITICS HPERTENSION |
Frequency |
Percent |
|
NO |
30 |
20.7 |
|
YES |
115 |
79.3 |
|
Total |
145 |
100.0 |
|
CO-MORBIDITICS LUNG DISEASE |
Frequency |
Percent |
|
NO |
139 |
95.9 |
|
YES |
6 |
4.1 |
|
Total |
145 |
100.0 |
|
CO-MORBIDITICS RENAL DISEASE |
Frequency |
Percent |
|
NO |
142 |
97.9 |
|
YES |
3 |
2.1 |
|
Total |
145 |
100.0 |
|
CO-MORBIDITICS HEART DISEASE |
Frequency |
Percent |
|
NO |
136 |
93.8 |
|
YES |
9 |
6.2 |
|
Total |
145 |
100.0 |
|
Co-morbidities, n (%) |
Patients received antibiotic < 1> |
Patients received antibiotic > 1 hour |
P value |
|
Hypertension |
70.0% |
84.2% |
0.045 |
|
Diabetes |
48.0% |
49.5% |
0.866 |
|
Renal disease |
2.0% |
2.1% |
0.966 |
|
Lung disease |
8.0% |
2.1% |
0.090 |
|
Heart disease |
4.0% |
7.4% |
0.424 |
Table2: Co-morbidities data for hypertension, renal, lung, heart diseases.
Cause of Admission in Icu Department
During our study period, the hospital had 145 cases from July 22, 2022 till November 28, 2022 in that female 63, 43.4% and male 82, 56.6%. The cause of admission of patient was maximum was for respiratory and lung disease 30, 2.1% kidney disease 17, 11.9% cardiovascular disease 16, 11.2% fever 15,10.5%, sepsis 14,9.8%. (Figure 3) There was no significant difference in the cause of admission in ICU of the patients as the p value is > 0.05 P value is 0.414. (Table 3)
|
Cause of ad- mission |
Frequency |
Percent |
P value |
|
FEVER |
15 |
10.5 |
0.414 |
|
RESPIRATORY AND LUNG DISEASE |
30 |
21 |
|
|
CARDIOVAS- CULAR DIS- EASE |
16 |
11.2 |
|
|
KIDNEY DIS- EASE |
17 |
11.9 |
|
|
DENGUE |
7 |
4.9 |
|
|
SEIZURE |
11 |
7.7 |
|
|
HERNIA |
3 |
2.1 |
|
|
JAUNDICE |
1 |
0.7 |
|
|
MALARIA |
4 |
2.8 |
|
|
SEPSIS |
14 |
9.8 |
|
|
ANEMIA |
2 |
1.4 |
|
|
COLD |
1 |
0.7 |
|
|
HEMORRHAG- IC STROKE |
2 |
1.4 |
|
|
HEPATIC ENCEPHALOP- ATHY |
3 |
2.1 |
|
|
GASTROIN- TESTINAL DISEASE |
9 |
6.3 |
|
|
HEMATEMESIS |
2 |
1.4 |
|
|
POLYTRAUMA |
1 |
0.7 |
|
|
LOOSE MO- TION |
1 |
0.7 |
|
|
PANCREATIC CANCER |
2 |
1.4 |
|
|
PANCYTOPE- NIA |
1 |
0.7 |
|
|
TRANSURE- THRAL RESEC- TION OF THE PROSTATE |
1 |
0.7 |
|
|
BREST CARCI- NOMA |
2 |
1.4 |
|
|
TOTAL |
145 |
100 |
Table3: Cause of admission in ICU Department

Figure3: distribution of Cause of admission In ICU department
Antibiotic received in the ICU during the ICU stay:
The patient in ICU are grouped in two parts, one who has received antibiotic within one hour and second group is patients who received antibiotic more than one hour. Antibiotic meropenem 67, 46.2% piperacillin & tazobactam 40, 27.6%, ceftazidime and avibactam 5,3.4%. (Figure 4) There was no significant difference in the source of admission in ICU of the patients as the p value is > 0.05 but Ceftazidime and Avibactam antibiotic P value is 0.029 which is significant difference. (Table 4.)

Figure 3: Antibiotic received in the ICU during the ITU stay
|
Antibiotics, n (%) |
Patients re- ceived antibi- otic < 1 hour |
Patients re- ceived antibi- otic > 1 hour |
P value |
|
Amoxicillin And Clavu- lanic Acid |
2.0% |
2.1% |
0.966 |
|
Amoxicil- lin-Potassi- um Clavula- nate |
2.0% |
3.2% |
0.686 |
|
Meropenem |
54.0% |
42.1% |
0.172 |
|
Piperacillin & Tazobactam |
22.0% |
30.5% |
0.275 |
|
Rifaximin |
0.0% |
1.1% |
0.467 |
|
Cephalospo- rin |
0.0% |
2.1% |
0.302 |
|
Cefopera- zone And Sulbactam |
2.0% |
0.0% |
0.167 |
|
Ceftazidime And Avibac- tam |
8.0% |
1.1% |
0.029 |
|
Azithromycin |
2.0% |
1.1% |
0.642 |
|
Ceftriaxone |
2.0% |
6.3% |
0.249 |
|
Doxycycline |
0.0% |
2.1% |
0.302 |
|
Carbapenem |
4.0% |
1.1% |
0.236 |
|
Teicoplanin |
8.0% |
11.6% |
0.501 |
|
Use of antibiotics within 1 hour of Admission |
Fre- quen- cy |
Per- cent |
Use of antibiotics within 1 hour of Ad- mission |
Fre- quen- cy |
Per- cent |
|
1.0 |
50 |
34.5 |
10.0 |
1 |
.7 |
|
2.0 |
36 |
24.8 |
11.0 |
1 |
.7 |
|
3.0 |
13 |
9.0 |
12.0 |
1 |
.7 |
|
4.0 |
4 |
2.8 |
13.0 |
1 |
.7 |
|
5.0 |
12 |
8.3 |
15.0 |
1 |
.7 |
|
6.0 |
10 |
6.9 |
16.0 |
1 |
.7 |
|
7.0 |
7 |
4.8 |
20.0 |
1 |
.7 |
|
8.0 |
2 |
1.4 |
|
|
|
|
9.0 |
4 |
2.8 |
Total |
145 |
100 |
Table4: Showing Antibiotic received and Timing in the ICU during the ICU stay
The Primary Outcomes Characteristics of the Study Population
Patients received antibiotic within 1hour of admission is 50 out of 145 patient its 34.5%. ITU outcome is divided into three different part one is ITU discharge 132, 97.9% ITU Death3, 2.1%. Hospital outcome of the patients is group into two variable that is death and discharge among 145 cases 11, 7.6% death and 134,92.4% discharges from hospital. (Figure 4) There was no significant difference in the primary outcome in ICU of the patients as the p value is > 0.05 but in ITU stay, P value is 0.002 and total stay, P value is 0.004 which is significant difference. (Table 5 & Table 6)

Figure4: The hospital outcome
|
Characteris- tics |
Patients re- ceived antibi- otic < 1 hour |
Patients re- ceived anti- biotic > 1 hour |
P value |
|
ITU stay, mean ± SD |
3.82 ± 2.73 |
5.03 ± 3.31 |
0.002 |
|
Number of day on mechanical ventilation mean ± SD |
0.02 ± 0.15 |
0.02 ± 0.21 |
0.623 |
|
Hospital stays, mean ± SD |
2.84 ± 3.03 |
3.26 ± 3.28 |
0.120 |
|
Total stay, mean ± SD |
6.66 ± 5.04 |
8.29 ± 5.29 |
0.004 |
|
ITU Outcome |
Frequency |
Percent |
|
ITU Discharge |
132 |
97.9 |
|
ITU Death |
3 |
2.1 |
|
Total |
145 |
100.0 |
|
Hospital Outcome |
Frequency |
Percent |
|
Death |
11 |
7.6 |
|
Discharge |
134 |
92.4 |
|
Total |
145 |
100.0 |
Table 5: Showing the Primary outcomes characteristics of the study population.
|
Hospital Outcome |
Patients re- ceived antibi- otic < 1 hour |
Patients re- ceived antibi- otic > 1 hour |
P value |
|
Death, n% |
4.0% |
9.5% |
0.237 |
|
Discharge, n% |
96.0% |
90.5% |
|
Table 6: Showing the hospital outcome
Mortality and Appropriate Antibiotics
Mortality of the patients were divided into two groups, antibiotic received within one hour and other major group is antibiotic received more than one hour to death. The antibiotic received within one hour is sub group into antibiotic (death 2,4.0% and discharge 48,96.0%) and antibiotic received within more than one hour antibiotic (10,10.5% death and 85,89.5% discharge) the p value is 0.175. There was no significant difference in the mortality and in choice of antibiotic in ICU of the patients as the p value is > 0.05 (Table 8)
Antibiotics was compared with antibiotic police it complies with the antibiotic drug list provide with the list with organism (Document: AMRI/DHK/HIC/POL/02 effective 15/09/2022) all are appropriate so we can conclude that antibiotic which were received against the Organism Antibiotic susceptibility (Table 8). Mortality total death 12 ,8.3%, discharge 133,91.7%. (Table 7). In the total population N=145 appropriate antibiotics received 48, 96.0%.
|
Mortality |
Frequency |
Percent |
|
Death |
12 |
8.3 |
|
Discharge |
133 |
91.7 |
|
Total |
145 |
100.0 |
Table 7: Showing the Mortality
|
Blood- stream Infections |
Number |
Antibiotic sus- ceptibility as per antibiotic police |
Antibiotics received (n) |
|
Escherich- ia coli |
7 |
Amoxy-clav Cef- triaxone, Pip-taz, Imipemen/Mero- penem, Ertapenem Amikacin, Colistin. |
Meropenem (5) Ceftriax- one (2) |
|
Klebsiella pneumo- niae |
2 |
Amoxy-clav, cef- triaxone, Pip-taz Imipenam/Mero- penam Ertapenem 90%, Amikacin, Colistin. |
Meropenam (2) |
|
Proteus mirabilis |
2 |
Amoxy-clav, Ceftri- axone, Ciprofloxa- cilin, Azithromycin. |
Azithromycin (2) |
|
Pseudo- monas aerugino- sa |
6 |
Ceftazidi- me,Pip-taz, Cefo- sulb Imipenam/ Meropenam, Amik- acin, Ciprofloxacin |
Meropenam (4) Ciproflox- acin (2) |
|
Staphy- lococcus aureus |
6 |
Penicillin, Vanco- mycin / Tecico- planin / Linezolid, Daptomycin, Cipro |
Piperacillin & Tazobactam (6) |
|
Urinary Iso- lates-OPD |
Num- ber |
Antibiotic sus- ceptibility as per antibiotic policy |
Antibiotics Received |
|
Candida species |
2 |
Fluco, Vori, AmpB, Echinocandins |
Meropenem (5) & Ceftri- axone (2) |
|
Enterococ- cus faecalis |
1 |
Penicillin, Nitrofuran 35%, Vancomy- cin / Teicoplanin, Linezolid 100% |
Piperacillin &Tazobact- am (1) |
|
Escherichia coli |
21 |
Amoxy-clav, Cef- triaxone, Pip-taz, Imipemen / Mero- penem, Ertapenem, Amikacin, Nitrofu- rantoin, Co trimox- azole, Colistin, Fosfomycin |
Meropen- em (18), Ceftazidime and Avibac- tam (3) |
|
Klebsiella pneumoniae |
2 |
Amoxyclav, ceftri- axone, cefo- sulb, Pip-taz, Imipemen / Meropenem, Cefta- Avi Amikacin, Colistin. |
Piperacillin & Tazobact- am (2) |
|
Pseudomo- nas aerugi- nosa |
2 |
Ceftazidime, Pip- taz, Cefo-sulb, Imi- penam / Merope- nam / Doripenem, Amikacin, Cipro/ Levo, Colistin |
Meropenem (2) |
|
Respiratory Isolates- ipd |
Number |
Antibiotic susceptibility as per antibiotic police |
Antibiotics Received |
|
Acinitobacter baumannii |
4 |
Ceftazidime,Pip-taz, Imipenem /Meropenem 0 %, Amik- acin, Cipro/ Levo Minocycline, Colistin 100%. |
Meropenem (4) |
|
Aeromonas sobria |
2 |
Amoxy-clav 92%, Ceftriaxone 100%, Ciprofloxacilin 55%, Azithromycin 100% |
Azithromycin (2) |
|
Escherichia coli and Strep- tococcus pneumoniae |
1 |
Amoxy-clav, Ceftriaxone, Pip-taz, Imipemen / Meropen- em, Ertapenem, Amikacin, Nitrofurantoin, Co-trimoxaz- ole,Colistin, Fosfomycin |
Meropenem (1) |
|
GRAM-NEGATIVE Bacilli |
3 |
Amoxyclav, ceftriaxone, cefo-sulb, Pip-taz, Imipemen / Meropenem, Cefta-Avi Amikacin, Colistin. |
Meropenem (1) |
|
PROTEUS MIRABILIS |
1 |
Amoxy-clav, Ceftriaxone, Ciprofloxacilin, Azithromycin |
Cephalosporin (1) |
Table 8: Showing the appropriate antibiotic as per the antibiotic police and list of antibiotic susceptibility (Document: AMRI/DHK/HIC/POL/02 effective 15/09/2022)
|
|
Antibiotics |
Total |
P- value |
|||
|
Within one hour |
More than one hour |
|||||
|
Mortality |
Death |
Count |
2 |
10 |
12 |
0.175 |
|
% within ANTIBIOTIC |
4.0% |
10.5% |
8.3% |
|||
|
Discharge |
Count |
48 |
85 |
133 |
||
|
% within ANTIBIOTIC |
96.0% |
89.5% |
91.7% |
|||
|
Total |
Count |
50 |
95 |
145 |
||
|
% within ANTIBIOTIC |
100.0% |
100.0% |
100.0% |
|||
Table 9: Showing the antibiotic Timing
Bacteriology and Culture Report
The blood culture was done for 97, 66.7% Escherichia coli 7,4.8% Pesudomonas aeruginosa 6,4.1%. The urine culture was done for 142 ,97.9% Escherichia coli 21, 14.5%, sputum culture 17,11.7% Acinetobacter baumanni4, 2.8%gram negative bacilli 3,2.1%, normal oral flora 3,2.1%. There was no significant difference in the culture report in ICU of the patients as the p value is > 0.05 but in blood culture p value is 0.045 which is significant difference. P value of urine culture is 0.00 and P value for sputum culture is 0.278.
|
Blood Culture Result |
Frequency |
Percent |
|
Escherichia coli |
7 |
4.8 |
|
Klebsiella pneumoniae |
2 |
1.4 |
|
Proteus mirabilis |
2 |
1.4 |
|
Pseudomonas aeruginosa |
6 |
4.1 |
|
Staphylococcus aureus |
4 |
2.8 |
|
Swab GRAM positive |
2 |
1.4 |
|
No Growth |
122 |
84.1 |
|
Total |
145 |
100.0 |
|
Sputum Culture Result |
Frequency |
Percent |
|
Aeromonas sobria |
2 |
1.4 |
|
Escherichia coli and Strep- tococcus pneumoniae |
1 |
.7 |
|
Gram-negative Bacilli |
3 |
2.1 |
|
Normal oral Flora |
3 |
2.1 |
|
Proteus mirabilis |
1 |
.7 |
|
No Growth |
131 |
90.4 |
|
Total |
145 |
100.0 |
|
Urine Culture Result |
Frequency |
Percent |
|
Candida species |
2 |
1.4 |
|
Enterococcus faecalis |
1 |
.7 |
|
Escherichia coli |
21 |
14.5 |
|
Gram negative bacteria |
2 |
1.4 |
|
Klebsiella pneumoniae |
2 |
1.4 |
|
Pseudomonas aeruginosa |
2 |
1.4 |
|
Streptococcus pneumoniae |
1 |
.7 |
|
No growth |
114 |
78.7 |
|
Total |
145 |
100.0 |
|
Characteristics |
Patients received antibi- otic < 1 hour (n%) |
Patients received antibiotic > 1 hour (n%) |
P value |
|
Blood Culture Result |
|
|
|
|
Escherichia coli |
8.0% |
3.2% |
0.045 |
|
Klebsiella pneumoniae |
4.0% |
0.0% |
|
|
Proteus mirabilis |
2.0% |
1.1% |
|
|
Pseudomonas aeruginosa |
8.0% |
2.1% |
|
|
Staphylococcus aureus |
4.0% |
2.1% |
|
|
No Growth |
72% |
90.6% |
|
|
Urine Culture Result |
|
|
|
|
Candida species |
4.0% |
0.0% |
0.00 |
|
Enterococcus faecalis |
0.0% |
1.0% |
|
|
Escherichia coli |
30.0% |
6.3% |
|
|
Klebsiella pneumoniae |
2.0% |
1.1% |
|
|
Pseudomonas aeruginosa |
0.0% |
1.1% |
|
|
Streptococcus pneumoniae |
0.0% |
1.1% |
|
|
No Growth |
60% |
88.5% |
|
|
Sputum Culture Result |
|
|
|
|
Acinetobacter baumannii |
6.0% |
1.1% |
0.278 |
|
Aeromonas sobria |
2.0% |
1.1% |
|
|
Escherichia coli and streptococcus pneumoniae |
0.0% |
1.1% |
|
|
Gram-negative bacilli |
2.0% |
2.1% |
|
|
Normal oral flora |
4.0% |
1.1% |
|
|
Proteus mirabilis |
2.0% |
0.0% |
|
|
No Growth |
84.0% |
93.7% |
|
Table 10: Showing the Bacteriology and culture report of ICU patients
Discussion
In this prospective observational study patient with age greater then 18 years old were Included and patient from any department to ICU department cause of admission is independent of any disease, door-to-antibiotic time was not associated with in-hospital mortality. Sterling et al. (18) found no significant differences when comparing the antibiotic administration within 3 hours from ED triage and within 1 hour from septic shock recognition. Door-to-antibiotic time and in-hospital mortality were the main focuses of this study, which showed that each extra hour (relative to door-to-antibiotic time ≤1 hour) was not associated with an increase in the mortality rate. The highest mortality rate in this study was in the door-to-antibiotic group of >1hours. Likewise, Peltan et al. (22) found that a door-to-antibiotic time cutoff of 3 hours was associated with mortality, but a cutoff of 1 hour did not show statistical significance. When the door-to-antibiotic times of ≤1 hour and >1hour were compared, the ≤1-hour group had greater severity of illnesses based on the ESI level and NEWS. For this reason, the door-to-antibiotic time of ≤1 hour had a higher mortality rate than the patients who received antibiotics later. The SSC guideline recommends antibiotic initiation within 1 hour. Nonetheless, many studies showed failure to achieve that goal. For instance, Abe et al. (23) found that 30.5% of cases received antibiotics within 1 hour. Ko et al. (24) revealed that the 1-hour target was achieved in 28.6% of septic shock patients treated in the EU. In this study, 48, 96.0%. of the patients received antibiotics within 1 hour and treated in ICU department.
During our study period, the hospital had 145 cases from July 22, 2022 till November 28, 2022 in that female 63,43.4% and male 82, 56.6%. The patients age group was divided into 7 groups 31-40,41-50,51-60,61-70,71-80,81-90,91-100 in which 61-70 maximum patient were admitted in hospital 46,31.7% and minimum patient admitted in hospital age group is 91-100 in hospital 1,0.7%. The cause of admission of patient was maximum was for respiratory and lung disease 30,2.1% kidney disease 17, 11.9 In my study it is demonstrated that gram-negative bacteria remains the major pathogen as has been demonstrated in most ICUs in India. Mortality of the patients were grouped into two groups, antibiotic received within one hour and other major group is antibiotic received more than one hour. The antibiotic received within one hour is sub group into antibiotic (death 2,4.0% and discharge 48,96.0%) and antibiotic received within more than one hour antibiotic (10,10.5 Number of patient population is small. The Sensitive % was not calculated. The study says the impact of appropriate antibiotics within 1hour of patient’s admission and the culuture report takes 3 days so Antibiotics were compared with antibiotic policy it complies with the antibiotic drug list provide with the list with organism (Document: AMRI/DHK/HIC/POL/02 effective 15/09/2022) all are appropriate so we can conclude that antibiotic which were received against the Organism Antibiotic susceptibility.Conclusion
Limitations
References
- Aronin, Steven I., Peter Peduzzi, and Vincent J. Quagliarello. “Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing.” Annals of internal medicine 129, no. 11_Part_1 (1998): 862-869.
- Auburtin, Marc, Michel Wolff, Julien Charpentier, Emmanuelle Varon, Yves Le Tulzo, Christophe Girault, Ismaël Mohammedi et al. “Detrimental role of delayed antibiotic administration and penicillin-nonsusceptible strains in adult intensive care unit patients with pneumococcal meningitis: the PNEUMOREA prospective multicenter study.” Critical care medicine 34, no. 11 (2006): 2758-2765.
- Gaieski, David F., Mark E. Mikkelsen, Roger A. Band, Jesse M. Pines, Richard Massone, Frances F. Furia, Frances S. Shofer, and Munish Goyal. “Impact of time to antibiotics on survival in patients with severe sepsis or septic shock in whom early goal-directed therapy was initiated in the emergency department.” Critical care medicine 38, no. 4 (2010): 1045-1053.
- Garnacho-Montero, José, Antonio Gutiérrez-Pizarraya, Ana Escoresca-Ortega, Esperanza Fernández-Delgado, and José María López-Sánchez. “Adequate antibiotic therapy prior to ICU admission in patients with severe sepsis and septic shock reduces hospital mortality.” Critical care 19, no. 1 (2015): 302.
- Anand Kumar, Anand Kumar, D. Roberts, K. E. Wood, B. Light, J. E. Parrillo, Satendra Sharma Satendra Sharma, R. Suppes et al. “Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock.” (2006): 1589-1596.
- Lepur, Dragan, and Bruno Baršić. “Community-acquired bacterial meningitis in adults: antibiotic timing in disease course and outcome.” Infection 35, no. 4 (2007): 225-231.
- Levy, Mitchell M., R. Phillip Dellinger, Sean R. Townsend, Walter T. Linde-Zwirble, John C. Marshall, Julian Bion, Christa Schorr et al. “The Surviving Sepsis Campaign: results of an international guideline-based performance improvement program targeting severe sepsis.” Intensive care medicine 36, no. 2 (2010): 222-231.
- Lu, Cheng-Hsien, Chi-Ren Huang, Wen-Neng Chang, Chin- Jung Chang, Ben-Chung Cheng, Ping-Yu Lee, Mei-Wen Lin, and Hsueh-Wen Chang. “Community-acquired bacterial meningitis in adults: the epidemiology, timing of appropriate antimicrobial therapy, and prognostic factors.” Clinical neurology and neurosurgery 104, no. 4 (2002): 352-358.
- Miner, James R., William Heegaard, Andrew Mapes, and Michelle Biros. “Presentation, time to antibiotics, and mortality of patients with bacterial meningitis at an urban county medical center.” The Journal of emergency medicine 21, no. 4 (2001): 387-392.
- Pines, Jesse M. “Timing of antibiotics for acute, severe infections.” Emergency medicine clinics of North America 26, no. 2 (2008): 245-257.
- Pines, Jesse M., Joshua A. Isserman, and Patrick B. Hinfey. “The measurement of time to first antibiotic dose for pneumonia in the emergency department: a white paper and position statement prepared for the American Academy of Emergency Medicine.” The Journal of emergency medicine 37, no. 3 (2009): 335-340.
- Proulx, N., D. Frechette, B. Toye, J. Chan, and S. Kravcik. “Delays in the administration of antibiotics are associated with mortality from adult acute bacterial meningitis.” Qjm 98, no. 4 (2005): 291-298.
- Nazarian, Devorah J., Orin L. Eddy, Thomas W. Lukens, Scott D. Weingart, and Wyatt W. Decker. “Clinical policy: critical issues in the management of adult patients presenting to the emergency department with community-acquired pneumonia.” Annals of emergency medicine 54, no. 5 (2009): 704-731.
- Puskarich, Michael A., Stephen Trzeciak, Nathan I. Shapiro, Ryan C. Arnold, James M. Horton, Jonathan R. Studnek, Jeffrey A. Kline, Alan E. Jones, and Emergency Medicine Shock Research Network (EMSHOCKNET. “Association between timing of antibiotic administration and mortality from septic shock in patients treated with a quantitative resuscitation protocol.” Critical care medicine 39, no. 9 (2011): 2066-2071.
- Sterling, Sarah A., W. Ryan Miller, Jason Pryor, Michael A. Puskarich, and Alan E. Jones. “The impact of timing of antibiotics on outcomes in severe sepsis and septic shock: a systematic review and meta-analysis.” Critical care medicine 43, no. 9 (2015): 1907-1915.
- van Zanten, Arthur RH. “The golden hour of antibiotic administration in severe sepsis: avoid a false start striving for gold.” Critical care medicine 42, no. 8 (2014): 1931-1932.
- Villar, Julian, Joseph P. Clement, Jim Stotts, Daniel Linnen, David J. Rubin, David Thompson, Antonio Gomez, and Christopher Fee. “Many emergency department patients with severe sepsis and septic shock do not meet diagnostic criteria within 3 hours of arrival.” Annals of emergency medicine 64, no. 1 (2014): 48-54.
- Vilella, Antonia L., and Charles F. Seifert. “Timing and appropriateness of initial antibiotic therapy in newly presenting septic patients.” The American journal of emergency medicine 32, no. 1 (2014): 7-13.
- Nauclér, P., A. Huttner, C. H. Van Werkhoven, M. Singer, P. Tattevin, S. Einav, and Thomas Tängdén. “Impact of time to antibiotic therapy on clinical outcome in patients with bacterial infections in the emergency department: implications for antimicrobial stewardship.” Clinical Microbiology and Infection 27, no. 2 (2021): 175-181.
- Rational antibiotic utilization in selected pediatric conditions. Malaysian Health Technology Assessment Unit,2002.
- Gaur, Aditya H., and B. Keith English. “The judicious use of antibiotics— An investment towards optimized health care.” The Indian Journal of Pediatrics 73, no. 4 (2006): 343-350.
- WHO global strategy for containment of antimicrobial resistance. World Health Organization, 2001.
- Chambers H: Antimicrobial agents: General considerations. Hardman JG, Limbird LE: The pharmacological basis of therapeutics. New York: MacGraw-Hill, 2001, 1143-69.
- Okeke, Iruka N., Oladiipo A. Aboderin, Denis K. Byarugaba, Kayode K. Ojo, and Japheth A. Opintan. “Growing problem of multidrug-resistant enteric pathogens in Africa.” Emerging infectious diseases 13, no. 11 (2007): 1640.
- Archibald LK, Reiller LB: Clinical microbiology in developing countries. Emerg Infect Dis, 2001, 7, 302-4.
- French, G. L. “Clinical impact and relevance of antibiotic resistance.” Advanced drug delivery reviews 57, no. 10 (2005): 1514-1527.
- Davey P; Brown E; Fenelon L; et al: Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev, 2005, (4), CD003543.
- Lodise, Thomas P., Peggy S. McKinnon, Linda Swiderski, and Michael J. Rybak. “Outcomes analysis of delayed antibiotic treatment for hospital-acquired Staphylococcus aureus bacteremia.” Clinical infectious diseases 36, no. 11 (2003): 1418- 1423.
- Harbarth, Stephan, Jorge Garbino, Jérome Pugin, Jacques A. Romand, Daniel Lew, and Didier Pittet. “Inappropriate initial antimicrobial therapy and its effect on survival in a clinical trial of immunomodulating therapy for severe sepsis.” The American journal of medicine 115, no. 7 (2003): 529-535.
- Gonzales, Ralph, John G. Bartlett, Richard E. Besser, John M. Hickner, Jerome R. Hoffman, and Merle A. Sande. “Principles of appropriate antibiotic use for treatment of nonspecific upper respiratory tract infections in adults: background.” Annals of internal medicine 134, no. 6 (2001): 490-494.
- Adorka, Matthias, Mitonga Kabwebwe Honoré, Martie Lubbe, Jan Serfontein, and Kirk Allen. “The impact of appropriate antibiotic prescribing on treatment evaluation parameters.” Journal of public health in Africa 4, no. 1 (2013): e2.
- Antimicrobial resistance: Does stopping a course of antibiotics early lead to antibiotic resistance? World Health Organization, 2020
- Oakley-Hannibal, E., and H. Gowda. “G142 (P) Are antibiotics administered within one hour in suspected neonatal sepsis as per nice guidelines?.” (2015): A61-A62.
- Houck, Peter M., Dale W. Bratzler, Wato Nsa, Allen Ma, and John G. Bartlett. “Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community-acquired pneumonia.” Archives of internal medicine 164, no. 6 (2004): 637-644.
- Joo, Young Min, Minjung Kathy Chae, Sung Yeon Hwang, Sang-Chan Jin, Tae Rim Lee, Won Chul Cha, Ik Joon Jo et al. “Impact of timely antibiotic administration on outcomes in patients with severe sepsis and septic shock in the emergency department.” Clinical and Experimental Emergency Medicine 1, no. 1 (2014): 35.
- Buckman, Sara A., Isaiah R. Turnbull, and John E. Mazuski. “Empiric antibiotics for sepsis.” Surgical infections 19, no. 2 (2018): 147-154.
- Vilella, Antonia L., and Charles F. Seifert. “Timing and appropriateness of initial antibiotic therapy in newly presenting septic patients.” The American journal of emergency medicine 32, no. 1 (2014): 7-13.
- Baré, Marisa, Xavier Castells, Angel Garcia, Marta Riu, Mercè Comas, and Maria José Gil Egea. “Importance of appropriateness of empiric antibiotic therapy on clinical outcomes in intra-abdominal infections.” International journal of technology assessment in health care 22, no. 2 (2006): 242-248.
- Lancaster, Jason W., Kenneth R. Lawrence, Jeffrey J. Fong, Shira I. Doron, Erik Garpestad, Stan A. Nasraway, and John W. Devlin. “Impact of an institution-specific hospital-acquired pneumonia protocol on the appropriateness of antibiotic therapy and patient outcomes.” Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy 28, no. 7 (2008): 852-862.
- Charles, Pierre Emmanuel, Claire Tinel, Saber Barbar, Serge Aho, Sébastien Prin, Jean Marc Doise, Nils Olivier Olsson, Bernard Blettery, and Jean Pierre Quenot. “Procalcitonin kinetics within the first days of sepsis: relationship with the appropriateness of antibiotic therapy and the outcome.” Critical care 13, no. 2 (2009): R38.
- Stewart, Philip S., and J. William Costerton. “Antibiotic resistance of bacteria in biofilms.” The lancet 358, no. 9276 (2001): 135-138.
- Capp, Roberta, Yuchiao Chang, and David FM Brown. “Effective antibiotic treatment prescribed by emergency physicians in patients admitted to the intensive care unit with severe sepsis or septic shock: where is the gap?.” The Journal of emergency medicine 41, no. 6 (2011): 573-580.
- Leekha, Surbhi, Christine L. Terrell, and Randall S. Edson. “General principles of antimicrobial therapy.” In Mayo clinic proceedings, vol. 86, no. 2, pp. 156-167. Elsevier, 2011.
- Christopher J. Grace, MD, FACP; Brad Robinson, MPH, MD: Empiric Antibiotic Selection. Infectious Disease Antimicrobial agents.
- Antibiotic Resistance. World Health Organization. 2020.
- Antibiotic police (Document: AMRI/DHK/HIC/POL/02 effective 15/09/2022)


