Archive : Article / Volume 2, Issue 1
- Research Article | DOI:
- https://doi.org/10.58489/2836-2187/009
A Narrative Review of Alterations in PANDAS Syndrome: Where Do We Draw the Line?
1Department of Medical Laboratory Sciences, Varastegan Institute for Medical Sciences, Mashhad, Iran
2Department of Microbiology, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
3Student Research Committee, Faculty of Medicine, Mashhad University of Medical Sciences, Iran
4Antimicrobial Resistance Research Centre, Mashhad University of Medical Sciences, Mashhad, Iran
5Department of Microbiology and Virology, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
Sepideh Hasanzadeh, Department of Microbiology and virology, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
Shaghayegh Zahmatkesh Moghadam, Behnam Azari, Davoud Mansouri, Ali Mehri, Kiarash Ghazvini, Sepideh Hasanzadeh, A Narrative Review of Alterations in PANDAS Syndrome: Where Do We Draw the Line? J. Microb. Res. Vol 2, Iss 1. (2023). DOI: 10.58489/2836-2187/009
© 2023 Sepideh Hasanzadeh, this is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Received Date: 01-01-2023
- Accepted Date: 12-01-2023
- Published Date: 18-01-2023
PANDAS/PANS, Obsessive-Compulsive Disorder, Tic disorder, Group A Streptococcus, Gut Microbiota
Abstract
PANDAS or Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection is a concerning disorder in childhood. Co-existence of the OCD and tic disorders after the infection of group A β-hemolytic streptococcus may cause PANDAS. PANDAS supported molecular mimicry as the cause of disorder. Characteristic changes that may be related to the alterations in the nervous system are demonstrated to be the first line of diagnosis. In PANDAS, some parts of the brain may undergo changes like dysfunction in basal ganglia and inflammation in striatum, etc. In this disorder not only do the symptoms tangle the nervous system, but also alterations in gut microbiota which correlate with the brain in psychiatric diseases are feasible. In addition, the studies on genetic aspects of PANDAS showed that changes can lead to the exacerbation in PANDAS. In this paper, we provide data related to signs and symptoms of PANDAS to clarify the importance of this disorder in childhood and adulthood as a side effect of streptococcal infection.
Highlights
- Presence of Autoantibodies against group A beta-hemolytic streptococcal antigen
- The acute onset of PANDAS in childhood and adolescence
- The impact of PANDAS on behavior, the nervous system, the gut microbiota, etc
- Effects of genetics on the severity of signs and symptoms of PANDAS
Introduction
The term Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection or PANDAS is a contentious topic that many details of it are still unclear [1, 2]. It is defined as part of a larger group called PANS or Pediatric Acute-onset Neuropsychiatric Syndrome [2]. The first description of this rare mental disorder was presented by Swedo et al. in 1998, based on 50 children with a sudden onset of obsessive-compulsive disorder (OCD) and tics [3]. Epidemiologically, PANDAS is more common in men [2.6:1] and usually occurs in childhood, but a minor percentage of cases show that PANDAS can happen after puberty [4, 5]. In recent years, scientists have suggested a connection between group A β-hemolytic streptococcus (GABHS) infections and subsequent development of OCD and tic disorders in children, the actual difference between PANS and PANDAS [1]. Consequently, there are some non-streptococcal infections such as Bornavirus or Mycoplasma pneumoniae that reveal the symptoms similar to those in PANDAS [6-8].
The hypothesis about the pathophysiology of PANDAS is molecular mimicry which involves antibodies produced against streptococcal antigens that can cross-react with brain proteins, particularly in the basal ganglia [3]. These antineuronal antibodies include anti-pyruvate kinase, anti-dopamine receptor, anti-Tubulin, and anti-lysoganglioside GM1, plus the receptors that antibodies cross-react with, including N-acetyl-β-D-glucosamine, lysoganglioside, and group A carbohydrate epitopes, and receptors of Dopamine (D1 and D2L) [2, 4, 5].
Neuroimmune interactions play a significant role in the pathophysiology of a variety of neuropsychiatric illnesses [1]. For instance, Both OCD and Tourette syndrome occur with alterations in the Cortico-basal ganglia circuitry [1, 9]. Although PANDAS is similar to a mental disorder called Sydenham chorea (SC), there are differences in the age of conflict and Systemic involvement [2].
As of 2017, the diagnostic criteria for PANDAS comprise five sections [4, 5]:
- Presence of OCD and/or tic disorders (chronic motor or vocal tic disorder)
- The onset of OCD and tics in childhood (between the age of 3 years old and period of puberty)
- Association with group A β-hemolytic streptococcus (GABHS) infection
- Co-existing with neurological abnormalities such as attention deficit hyperactivity disorder (ADHD), choreiform movements, etc.
- Sudden onset and episodic course of symptoms
In addition, the state of Streptococcus pyogenes infection is analyzed and measured by the antibody titers of ASO and/or anti-DNase B in the blood sample and a positive throat culture of GABHS [3-5]. However, as a result of 2019, the increased number in specific antibodies of Streptococcus pyogenes is more accurate than a positive culture and titer evaluations because these are some how misleading ways to a carrier state [10]. Serological methods consider the possibility of the carrier state [3]. One of the more convenient methods to diagnose PANDAS is Cunningham Panel. Based on auto-antibody levels, it has shown an overall of 90% ac- curacy, a sensitivity of 88%, and a specificity of 92% [11]. When evaluated by the changes in the number of positive tests, the overall accuracy of the Cunningham panel drops to 86% and its sensitivity, and specificity decreases to 88% and 83% respectively [11].
Regarding PANDAS exacerbation, Swedo et al. described six months between a GABHS infection and onset of OCD [12]. Nevertheless, studies showed that the cut-off periods vary between 4 and 22 weeks [3]. The worsening of PANDAS is 30% due to GABHS infection, 20% because of non-streptococcal illnesses, and 50% because of unknown factors [13]. PANDAS has profound effects not only on the central nervous system but also on gut microbiota, general behavior, remission period, and genetics (Table 1). Some factors correlate with the PANDAS symptoms such as upper respiratory infections, the age of onset, neurological impairments, family history of autoimmune disorder or diseases that are associated with GABHS like rheumatic fever [14]. The purpose of this paper is to review and summarize available articles about the signs and symptoms of PANDAS to show how crucial this condition is in childhood and adulthood.
| Category of Symptoms | Signs and Symptoms | References |
| Characteristic Symptoms* | tics, OCD, motor hyperactivity, urinary urgency such as enuresis, impulsivity, anxiety, eating disorder, hallucination, insomnia, dilated pupils, aggressive- ness, depression, and choreiform movements | (7, 14, 15) |
| Alterations in Nervous system | Basal ganglia | -4 |
| Striatum and Striatal Interneurons | ||
| Microglia | ||
| Alterations in Gut Microbiota | Higher level: Bacteroidetes, Rikenellaceae and Odoribacteriaceae | -4 |
| Lower level: Firmicutes and Actinobacteria | ||
| Total absence: Saccharibacteria and Turicibacteraceae, Tissierellaceae, Gemellaceae, and Carnobacteriaceae, Corynebacteriaceae and Lachnospiraceae | ||
| Genetic correlation with PANDAS | Variant in exon 1 of MBL2, polymorphism of codon 54 of MBL2, TNF-α -308 | (4, 16-18) |
| AA polymorphism, the expression of SLITRK1, and epigenetic changes | ||
| *Behavioral-cognitive therapy and SSRIs are available to treat Characteristic Symptoms. | ||
Table1: Summary of Signs and Symptoms of PANDAS Syndrome
Signs and Symptoms of PANDAS
GABHS and the Symptoms of Related Diseases
GABHS is responsible for the majority of diseases in children like pharyngitis and rheumatic fever [15]. Streptococcal pharyngitis, difficulty in swallowing, nausea and vomiting, fever, headache, and enlargement in anterior cervical lymph nodes [16,17]. Rheumatic fever is also an autoimmune disease that shows inflammation in joints, myocarditis, marginal erythema, subcutaneous nodules, and even incidents of SC [18, 19]. Indeed, 2%-4% of children with rheumatic fever are likely to develop OCD [4]. The occurrence of OCD may accompany SC [4]. SC is characterized by the following symptoms: unilateral involuntary movements in facial and limb muscles, emotional instability, irritability, and anxiety [20]. Besides the sore throat due to GABHS, some skin infections such as scarlet fever, lupus, petechiae, vasculitis, perianal redness are associated with the onset of PANDAS, too [21, 22].
Characteristic Symptoms
PANDAS is a complicated condition that affects most children’s lives. The main aspect of PANDAS cases is about characteristic changes associated with alteration in the nervous system. These symptoms may include tics, OCD, motor hyperactivity, urinary urgency such as enuresis, impulsivity, anxiety, hallucination, insomnia, dilated pupils, aggressiveness, depression, and choreiform movements of fingers and toes which mainly appear in Huntington disease [14, 23, 24]. The symptoms impact school functioning of children occurred rapidly, between 24 to 72 hours, and characterized by math difficulties, low processing, fatigue, dysgraphia, difficulty with memorizing, and lack of concentration [4, 28]. It is necessary to mention that in PANDAS, the characteristic symptoms such as anxiety manifest more severe than somatic symptoms such as joint pain [29]. The rapid onset of these symptoms makes it hard to clarify the difference between OCD and PANDAS [30].
Alterations in Nervous System
Basal ganglia belong to a group of subcortical nuclei embedded in the brain hemispheres responsible for motor and mental control [31]. Disruption in this part forms several movement disorders such as parkinsonian syndrome, dystonia, and tics [31]. The Striatum, a part of basal ganglia, is essential for voluntary motor control [32]. Moreover, Striatum involves neurons that signal our social action [32]. On the other side, Microglia are known as resident immune cells located in the central nervous system (CNS) [1, 33]. Their activation is associated with releasing reactive oxygen products and inflammatory molecules [33]. There is evidence that Microglia have novel roles in neural development, phagocytize dying cells, promote programmed cell death, regulating syn- apse development, homeostasis, brain plasticity, and neurogenesis [1, 33-35]. PANDAS is identified by impairments in the central or peripheral nervous system, or both and particularly in basal ganglia, Striatum and Striatal Interneurons, Microglia, etc. [4].
Basal Ganglia
There is evidence that monoclonal antibodies attend to signal neuronal cells and stimulate them to release dopamine and activate calcium-calmodulin dependent protein (CaM) kinase II in basal ganglia [4, 36]. This activation is associated with the progression of the choreic movements [4]. The OCD and tics show abnormalities in cortico-basal ganglia circuitry [37]. The autoantibodies against M18 strain of Streptococcus pyogenes bind to the receptors in basal ganglia [38]. And also, the presence of anti-basal ganglia antibodies in affected individuals’ serum is a marker with high sensitivity and specificity [39]. Epidemiologically, two-third of PANDAS patients show these antibodies [40]. On one hand, the corpus striatum and basal ganglia are enlarged due to an increased number of antineuronal antibody titers [41]. On the other hand, it is clear that the gray matter of basal ganglia had a greater volume, although the white matter shows a reduced amount (based on Cabrera et al.) [42]. Nevertheless, the size of related structures such as caudate, putamen, and globus pallidus increased while the size of thalamus and cerebrum stayed the same (Figure 1) [43]. Surprisingly, the severity of symptoms doesn’t correlate with the enlargement of basal ganglia [4]. The variations within basal ganglia volumes are recognizable whereas there is no evidence about the changes in basal ganglia [4].
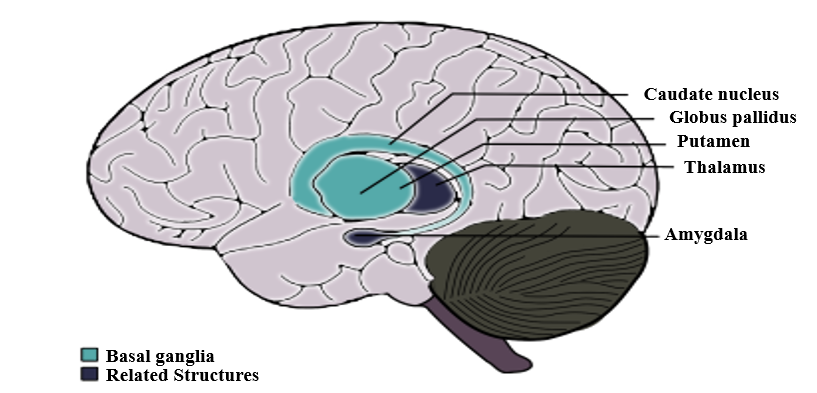
Figure1: Alterations in the nervous system is not just limited to basal ganglia although the related structures may undergo changes due to the presence autoantibodies.
Striatum and Striatal Interneurons
As Giedd et al. reported and by PET imaging, the enlargement in the striatum in PANDAS and SC patients is observable [37, 43]. There is a close interaction between the dysregulation within striatal neurons and PANDAS behavioral symptoms and it plays a significant role in tic disorders [37, 44]. More recently, the striatum may be inflamed in PANDAS and Tourette syndrome [37]. However, the inflammation is higher through the bilateral caudate and lentiform nucleus [1, 4]. The pathophysiology of tic disorder and Tourette syndrome is associated with cholinergic interneurons [45]. Furthermore, the cholinergic interneurons express dopamine receptors, which is a building-up situation for autoantibodies to bind in patients [4, 37].
Microglia
The CD68+/Iba1+ activated microglia in the olfactory bulb increases due to frequent internasal GABHS infections [46]. However, it has shown that microglia are beneficial to present the GABHS antigens to immune cells such as Th17 [1, 47]. As mentioned before, microglia are necessary for brain development and its regular action by synaptic pruning [1]. An animal experiment showed that synaptic pruning will increase in PANDAS [47]. Fractalkine/fractalkine receptor signaling pathways may disrupt in PANDAS and result in neural and behavioral changes [48].
Alterations in Gut Microbiota
Gut microbiome includes anaerobic bacteria, fungi, parasites, and viruses that are significant as a part of every mammalian immune system and play a critical role in adult development and homeostasis [49, 50]. If the homeostasis of Gastrointestinal altered, the development and function of the nervous system would considerably be affected (Figure 2) [50, 51]. For instance, the brain-derived neurotrophic factor (BDNF) levels can be affected by gut microbiota and bacterial dysbiosis may produce systemic inflammation [49, 52]. The axis develops during the intrauterine period [4]. The antimicrobial treatment during pregnancy, vaccination, exposure to chemical or other microorganisms, types of childbirth, etc. may affect this axis [53, 54]. Indeed, some disorders prove the close interaction such as Parkinson's disease, Alzheimer's disease, schizophrenia, multiple sclerosis, anxiety, autism, anorexia nervosa, ADHD, alcohol dependence, bipolar disorders, and migraine pain [4, 49]. The bacterial commensals that produce γ-aminobutyric acid in the gastrointestinal tract may induce neuropsychiatric symptoms, too [4]. It is concluded that the changes in gut microbiota can lead us to enhance PANDAS development.
In his studies about the interactions between human microbiota, gut, and brain, Quagliariello et al. found that the amount of some bacterial families may change due to PANDAS (Table 2) [55]. The group of 4-8 years old patients presented the increased level of Bacteroidaceae, Rikenellaceae, and Odoribacteriaceae [4]. Contrariwise, the level of Firmicutes and Actinobacteria were lower than expected in this group of patients [4]. Also, TM7 phylum or Saccharibacteria, and some of the Firmicutes families such as Turicibacteraceae, Tissierellaceae, Gemellaceae, Carnobacteriaceae Corynebacteriaceae, and Lachnospiracea were absent during the incidence of PANDAS [4]. Given the fact that the age of conflict is a crucial factor in the discrepancy between gut microbiota levels, the patients above 9 years old represent different results from the other group [4]. An increased level of Peptostreptococcaceae and Erysipelotrichaceae and lower levels of Rikenellaceae and Barnesiellacea were dramatically observable in these patients [4]. Furthermore, it is essential to mention that Bacteroidaceae presented as the most inclusive family of gut microbiota in PANDAS patients [4].
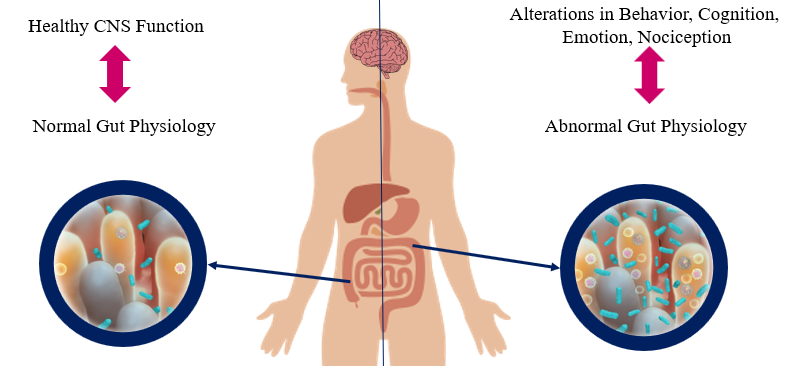
Figure2: The interaction of gut microbiota and the brain is mutual through the microbiota-gut-brain axis.
| Age | Higher level | Lower level | Total absence |
| 4-8 years old | Bacteroidaceae*, Rikenellaceae, and Odoribacteria- ceae | Firmicutes Actinobacteria | Saccharibacteria Some of the firmicutes include Turicibacteraceae, Tissierellaceae, Gemellaceae, and Carnobacteriaceae, Corynebacteriaceae and Lachnospiraceae |
| Above 9 years old | Peptostreptococcaceae and Erysipelotrichaceae | Rikenellaceae and Barnesi- ellacea | _ |
Table2: Summary of the discrepancy between gut microbiota
Genetic correlation with PANDAS
There are some genetic susceptibilities to psychiatric diseases like OCD, anxiety, attention deficit hyperactivity disorder (ADHD), pervasive developmental disorder (PDD), etc. [21]. According to what Murphy et al. mentioned, the rate of maternal autoimmune diseases increased in a group of patients with either OCD, tic disorders, or both [56]. The expression of the Trk-like family member 1 (SLITRK1) gene is associated with the cholinergic interneurons in the adult striatum [27]. On the other side, GABHS is highly susceptible to bind to Mannose-binding lectin (MBL), which is a critical part of immune responses [4]. Surprisingly, any changes like variants in exon 1 or polymorphism of codon 54 of MBL2 increases the probability of PANDAS [57]. Changes in TNF-α like, TNF-α-308 AA polymorphism, is associated with the exacerbation of PANDAS symptoms [26]. Since the incidence and severity of autoimmune disorders like PANDAS vary among identical siblings, we would have concluded that environmental factors mostly affect this discordance [25]. Moreover, prenatal and postnatal difficulties and early exposure to GABHS may affect the development of the immune system at the epigenetic level. Methylation of the DNA or modifications in the way histones package the DNA are examples of epigenetic changes [25].
Exacerbation in Remission Period
PANDAS treatment varies including antibiotic therapy (acute antibiotic therapy and prophylaxis) such as Penicillin V, Azithromycin, Amoxicillin-clavulanate, Cephalosporins, psychoactive drugs such as selective serotonin reuptake inhibitors (SSRIs) like fluoxetine, Escitalopram, Lorazepam, Nonsteroidal anti-inflammatory drugs (NSAIDs), Corticosteroid, Tonsillectomy, Intravenous immunoglobulin therapy (IVIG), Plasmapheresis, Vitamin D, and behavioral-cognitive therapy [4, 58, 59]. The remission period of PANDAS may take 3.3 years on average (based on Leon et al.) [13]. Unlike PANDAS, SC completes a remission in less than a year [60]. This exacerbation includes ADHD, oppositional defiant disorder (ODD), depression, enuresis, and dysthymia (Figure 3) [61]. After the remission of OCD and/or tic disorders, it is probable that children with PANDAS show signs of inattention, impulsiveness, and emotional lability, too [4]. Affected children also rarely encounter constant anxiety and fear as well as OCD, whereas other symptoms have disappeared [4].
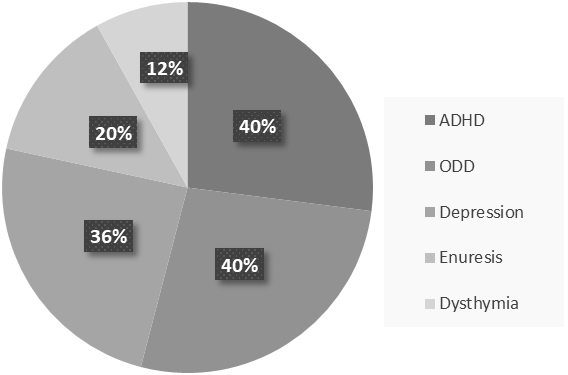
Figure3: According to the PANDAS cohorts, the percentages of psychiatric comorbidities from more common to less common has shown above.
Conclusion
Even though PANDAS was first introduced in 1998 by Swedo et al., it is still tough to distinguish its differences between a typical OCD or tic disorder [4]. PANDAS can be counted as a subgroup of PANS, characterized by restricted food intake after infection [2]. PANDAS has a high similarity to Sydenham chorea, which is another side effect of GABHS infection [62]. Environmental factors, coexisting diseases such as OCD, and genetic predispositions may be associated with this disease [63]. Experts define PANDAS by its characteristic symptoms ranging from typical anxiety to acute onset of OCD. The size of some parts of the brain like basal ganglia, striatum, caudate, and putamen is altered in PANDAS by binding autoantibodies in the brain. As seen in some cases, PANDAS may affect the gut microbiota, which in turn can exacerbate the onset of disease. Besides the signs and symptoms, the antibody titers of ASO or anti-DNase B in the blood sample and a positive throat culture of GABHS are helpful in the diagnosis of PANDAS [3-5]. Another method used these days is Cunningham Panel, which shows autoantibodies produced in PANDAS [9]. Although the variety of PANDAS treatments may take 3.3 years on average to cure children, it has proven that children may experience psychiatric symptoms in the remission period [48]. PANDAS has been the topic for many articles, but despite available studies, some aspects of it are still unclear.
Conflicts of Interest
None to declare.
Ethical Approval
Patient relative's consent was obtained for publication.
References
- Frick, Luciana, and Christopher Pittenger. “Microglial dysreg- ulation in OCD, Tourette syndrome, and PANDAS.” Journal of immunology research 2016, no. 1 (2016): 8606057.
- Gamucci, Alessandra, Sara Uccella, Lucia Sciarretta, Maria D’Apruzzo, Maria Grazia Calevo, Maria Margherita Mancardi, Edvige Veneselli, and Elisa De Grandis. “PANDAS and PANS: clinical, neuropsychological, and biological characterization of a monocentric series of patients and proposal for a diagnostic protocol.” Journal of child and adolescent psychopharmacolo- gy 29, no. 4 (2019): 305-312.
- Nielsen, Marie Ødum, Ole Köhler-Forsberg, Carsten Hjorthøj, Michael Eriksen Benros, Merete Nordentoft, and Sonja Orlovs- ka-Waast. “Streptococcal infections and exacerbations in PAN- DAS: a systematic review and meta-analysis.” The Pediatric infectious disease journal 38, no. 2 (2019): 189-194.
- Baj, Jacek, Elżbieta Sitarz, Alicja Forma, Katarzyna Wróblews- ka, and Hanna Karakuła-Juchnowicz. “Alterations in the ner- vous system and gut microbiota after β-hemolytic streptococ- cus group A infection—characteristics and diagnostic criteria of PANDAS recognition.” International journal of molecular sci- ences 21, no. 4 (2020): 1476.
- Kırık, Serkan, Olcay Güngör, and Yasemin Kırık. “Importance of Streptococci Infections in Childhood Neuropsychiatric Dis- orders.” Şişli Etfal Hastanesi tıp Bülteni 53, no. 4 (2019): 441.
- Dietrich, D. E., Y. Zhang, L. Bode, T. F. Münte, U. Hauser, P. Schmorl, C. Richter-Witte et al. “Brain potential amplitude var- ies as a function of Borna disease virus-specific immune com- plexes in obsessive–compulsive disorder.” Molecular psychia- try 10, no. 6 (2005): 519-520.
- Matsuo, Muneaki, Katsunori Tsuchiya, Yuhei Hamasaki, and Harvey S. Singer. “Restless legs syndrome: association with streptococcal or mycoplasma infection.” Pediatric neurology 31, no. 2 (2004): 119-121.
- Müller, Norbert, Michael Riedel, Christa Blendinger, Karin Oberle, Enno Jacobs, and Marianne Abele-Horn. “Mycoplasma pneumoniae infection and Tourette’s syndrome.” Psychiatry re- search 129, no. 2 (2004): 119-125.
- Leckman, James F., Michael H. Bloch, Megan E. Smith, Daouia Larabi, and Michelle Hampson. “Neurobiological substrates of Tourette’s disorder.” Journal of child and adolescent psycho- pharmacology 20, no. 4 (2010): 237-247.
- Johnson, Dwight R., Roger Kurlan, James Leckman, and Ed- ward L. Kaplan. “The human immune response to streptococ- cal extracellular antigens: clinical, diagnostic, and potential pathogenetic implications.” Clinical infectious diseases 50, no. 4 (2010): 481-490.
- Shimasaki, Craig, Richard E. Frye, Rosario Trifiletti, Michael Cooperstock, Gary Kaplan, Isaac Melamed, Rosalie Green- berg et al. “Evaluation of the Cunningham Panel™ in pedi- atric autoimmune neuropsychiatric disorder associated with streptococcal infection (PANDAS) and pediatric acute-onset neuropsychiatric syndrome (PANS): Changes in antineuronal antibody titers parallel changes in patient symptoms.” Journal of neuroimmunology 339 (2020): 577138.
- Swedo SE, Leonard HL, Garvey M, Mittleman B, Allen AJ, Perlmutter S, et al. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: clinical de- scription of the first 50 cases. American Journal of Psychiatry. 1998;155(2):264-71.
- Leon, Jill, Rebecca Hommer, Paul Grant, Cristan Farmer, Pre- cilla D’Souza, Riley Kessler, Kyle Williams, James F. Leckman, and Susan Swedo. “Longitudinal outcomes of children with pe- diatric autoimmune neuropsychiatric disorder associated with streptococcal infections (PANDAS).” European child & adoles- cent psychiatry 27 (2018): 637-643.
- Macerollo, Antonella, and Davide Martino. “Pediatric autoim- mune neuropsychiatric disorders associated with streptococcal infections (PANDAS): an evolving concept.” Tremor and other hyperkinetic movements 3 (2013): tre-03.
- Soderholm, Amelia T., Timothy C. Barnett, Matthew J. Sweet, and Mark J. Walker. “Group A streptococcal pharyngitis: Im- mune responses involved in bacterial clearance and GAS-as- sociated immunopathologies.” Journal of leukocyte biology 103, no. 2 (2018): 193-213.
- Arnold JC, Nizet V. Pharyngitis. Principles and Practice of Pe- diatric Infectious Diseases. 2012:199.
- Shaikh, Nader, Nithya Swaminathan, and Emma G. Hooper. “Accuracy and precision of the signs and symptoms of strepto- coccal pharyngitis in children: a systematic review.” The Jour- nal of pediatrics 160, no. 3 (2012): 487-493.
- Karthikeyan, Ganesan, and Luiza Guilherme. “Acute rheumatic fever.” The Lancet 392, no. 10142 (2018): 161-174.
- Webb RH, Grant C, Harnden A. Acute rheumatic fever. Bmj. 2015;351:h3443.
- Punukollu, Mallika, Nadine Mushet, Marisa Linney, Colm Hen- nessy, and Michael Morton. “Neuropsychiatric manifestations of Sydenham’s chorea: a systematic review.” Developmental Medicine & Child Neurology 58, no. 1 (2016): 16-28.
- Chang, Kiki, Jennifer Frankovich, Michael Cooperstock, Mad- eleine W. Cunningham, M. Elizabeth Latimer, Tanya K. Mur- phy, Mark Pasternack et al. “Clinical evaluation of youth with pediatric acute-onset neuropsychiatric syndrome (PANS): rec- ommendations from the 2013 PANS Consensus Conference.” Journal of Child and Adolescent Psychopharmacology 25, no. 1 (2015): 3-13.
- Carelli, Rosanna, and Stefano Pallanti. “Streptococcal infec- tions of skin and PANDAS.” Dermatologic Therapy 27, no. 1 (2014): 28-30.
- Swedo, Susan E., James F. Leckman, and Noel R. Rose. “From research subgroup to clinical syndrome: modifying the PANDAS criteria to describe PANS (pediatric acute-onset neu- ropsychiatric syndrome).” Pediatr Therapeut 2, no. 2 (2012): 113.
- Zibordi, Federica, Giovanna Zorzi, Miryam Carecchio, and Nardo Nardocci. “CANS: Childhood acute neuropsychiatric syndromes.” European Journal of Paediatric Neurology 22, no. 2 (2018): 316-320.
- Lewin, Adam B., Eric A. Storch, and Tanya K. Murphy. “Pedi- atric autoimmune neuropsychiatric disorders associated with Streptococcus in identical siblings.” Journal of child and ado- lescent psychopharmacology 21, no. 2 (2011): 177-182.
- Luleyap, H. Umit, Dilge Onatoglu, M. Bertan Yilmaz, Davut Alptekin, Aysegul Y. Tahiroglu, Salih Cetiner, Ayfer Pazarba- si, Ilker Unal, Ayse Avci, and Gamze Comertpay. “Association between pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections disease and tumor necrosis factor-α gene− 308 g/a,− 850 c/t polymorphisms in 4-12-year-old children in Adana/Turkey.” Indian Journal of Hu- man Genetics 19, no. 2 (2013): 196.
- O’Roak, B. J., T. M. Morgan, D. O. Fishman, E. Saus, P. Alon- so, M. Gratacos, X. Estivill et al. “Additional support for the association of SLITRK1 var321 and Tourette syndrome.” Mo- lecular psychiatry 15, no. 5 (2010): 447-450.
- Murphy, Tanya K., Roger Kurlan, and James Leckman. “The immunobiology of Tourette’s disorder, pediatric autoimmune neuropsychiatric disorders associated with Streptococcus, and related disorders: a way forward.” Journal of child and adoles- cent psychopharmacology 20, no. 4 (2010): 317-331.
- Calaprice, Denise, Janice Tona, Ellisa Carla Parker-Athill, and Tanya K. Murphy. “A survey of pediatric acute-onset neuropsy- chiatric syndrome characteristics and course.” Journal of Child and Adolescent Psychopharmacology 27, no. 7 (2017): 607- 618.
- Murphy, Tanya K., Diana M. Gerardi, and E. Carla Parker-Athill. “The PANDAS controversy: why (and how) is it still unsettled?.” Current Developmental Disorders Reports 1 (2014): 236-244.
- Lanciego, José L., Natasha Luquin, and José A. Obeso. “Func- tional neuroanatomy of the basal ganglia.” Cold Spring Harbor perspectives in medicine 2, no. 12 (2012): a009621.
- Báez-Mendoza, Raymundo, and Wolfram Schultz. “The role of the striatum in social behavior.” Frontiers in neuroscience 7 (2013): 233.
- Wake, Hiroaki, and R. Douglas Fields. “Physiological function of microglia.” Neuron glia biology 7, no. 1 (2011): 1-3.
- Du, Li, Ying Zhang, Yang Chen, Jie Zhu, Yi Yang, and Hong-Li- ang Zhang. “Role of microglia in neurological disorders and their potentials as a therapeutic target.” Molecular neurobiolo- gy 54, no. 10 (2017): 7567-7584.
- Tremblay, Marie-Ève, Beth Stevens, Amanda Sierra, Hiroaki Wake, Alain Bessis, and Axel Nimmerjahn. “The role of microg- lia in the healthy brain.” Journal of Neuroscience 31, no. 45 (2011): 16064-16069.
- da Rocha, Felipe Filardi, Humberto Correa, and Antonio Lucio Teixeira. “Obsessive–compulsive disorder and immunology: A review.” Progress in Neuro-Psychopharmacology and Biologi- cal Psychiatry 32, no. 5 (2008): 1139-1146.
- Frick, Luciana R., Maximiliano Rapanelli, Kantiya Jindachom- thong, Paul Grant, James F. Leckman, Susan Swedo, Kyle Williams, and Christopher Pittenger. “Differential binding of an- tibodies in PANDAS patients to cholinergic interneurons in the striatum.” Brain, behavior, and immunity 69 (2018): 304-311.
- Brimberg, Lior, Itai Benhar, Adita Mascaro-Blanco, Kathy Al- varez, Dafna Lotan, Christine Winter, Julia Klein et al. “Be- havioral, pharmacological, and immunological abnormalities after streptococcal exposure: a novel rat model of Sydenham chorea and related neuropsychiatric disorders.” Neuropsycho- pharmacology 37, no. 9 (2012): 2076-2087.
- Church, A. J., R. C. Dale, and G. Giovannoni. “Anti-basal gan- glia antibodies: a possible diagnostic utility in idiopathic move- ment disorders?.” Archives of Disease in Childhood 89, no. 7 (2004): 611-614.
- Pavone, Piero, Rio Bianchini, Enrico Parano, Gemma Incor- pora, Renata Rizzo, Luigi Mazzone, and Rosario R. Trifiletti. “Anti-brain antibodies in PANDAS versus uncomplicated strep- tococcal infection.” Pediatric neurology 30, no. 2 (2004): 107- 110.
- Giedd, J. N., J. L. Rapoport, M. J. P. Kruesi, C. Parker, M. B. Schapiro, A. J. Allen, H. L. Leonard et al. “Sydenham’s chorea: magnetic resonance imaging of the basal ganglia.” Neurology 45, no. 12 (1995): 2199-2202.
- Cabrera, Brenda, César Romero-Rebollar, Luis Jiménez-Ánge- les, Alma D. Genis-Mendoza, Julio Flores, Nuria Lanzagorta, María Arroyo et al. “Neuroanatomical features and its useful- ness in classification of patients with PANDAS.” CNS spec- trums 24, no. 5 (2019): 533-543.
- Giedd, Jay N., Judith L. Rapoport, Marjorie A. Garvey, Susan Perlmutter, and Susan E. Swedo. “MRI assessment of children with obsessive-compulsive disorder or tics associated with streptococcal infection.” American Journal of Psychiatry 157, no. 2 (2000): 281-283.
- Bonsi, Paola, Dario Cuomo, Giuseppina Martella, Graziella Madeo, Tommaso Schirinzi, Francesca Puglisi, Giulia Ponte- rio, and Antonio Pisani. “Centrality of striatal cholinergic trans- mission in basal ganglia function.” Frontiers in neuroanatomy 5 (2011): 6.
- Kataoka, Yuko, Paul SA Kalanithi, Heidi Grantz, Michael L. Schwartz, Clifford Saper, James F. Leckman, and Flora M. Vaccarino. “Decreased number of parvalbumin and cholinergic interneurons in the striatum of individuals with Tourette syn- drome.” Journal of Comparative Neurology 518, no. 3 (2010): 277-291.
- Dileepan, Thamotharampillai, Erica D. Smith, Daniel Know- land, Martin Hsu, Maryann Platt, Peter Bittner-Eddy, Brenda Cohen et al. “Group A Streptococcus intranasal infection pro- motes CNS infiltration by streptococcal-specific Th17 cells.” The Journal of clinical investigation 126, no. 1 (2016): 303-317.
- Tay, Tuan Leng, Catherine Béchade, Ivana D’andrea, Ma- rie-Kim St-Pierre, Mathilde S. Henry, Anne Roumier, and Marie-Eve Tremblay. “Microglia gone rogue: impacts on psy- chiatric disorders across the lifespan.” Frontiers in molecular neuroscience 10 (2018): 421.
- Zhan, Yang, Rosa C. Paolicelli, Francesco Sforazzini, Laetitia Weinhard, Giulia Bolasco, Francesca Pagani, Alexei L. Vys- sotski et al. “Deficient neuron-microglia signaling results in im- paired functional brain connectivity and social behavior.” Na- ture neuroscience 17, no. 3 (2014): 400-406.
- Galland, Leo. “The gut microbiome and the brain.” Journal of medicinal food 17, no. 12 (2014): 1261-1272.
- Zhu, Xiqun, Yong Han, Jing Du, Renzhong Liu, Ketao Jin, and Wei Yi. “Microbiota-gut-brain axis and the central nervous sys- tem.” Oncotarget 8, no. 32 (2017): 53829.
- Gawlik-Kotelnicka, Oliwia, and Michał Polguj. “Can microbiol- ogy affect psychiatry? A link between gut microbiota and psy- chiatric disorders.” Psychiatr. Pol 52, no. 6 (2018): 1023-1039.
- Sherwin, Eoin, Kiran V. Sandhu, Timothy G. Dinan, and John F. Cryan. “May the force be with you: the light and dark sides of the microbiota–gut–brain axis in neuropsychiatry.” CNS drugs 30, no. 11 (2016): 1019-1041.
- Jašarević, Eldin, Christopher D. Howard, Ana M. Misic, Daniel P. Beiting, and Tracy L. Bale. “Stress during pregnancy alters temporal and spatial dynamics of the maternal and offspring microbiome in a sex-specific manner.” Scientific Reports 7, no. 1 (2017): 44182.
- Milliken, Sarah, Ruridh M. Allen, and Ronald F. Lamont. “The role of antimicrobial treatment during pregnancy on the neo- natal gut microbiome and the development of atopy, asthma, allergy and obesity in childhood.” Expert opinion on drug safety 18, no. 3 (2019): 173-185.
- Quagliariello, Andrea, Federica Del Chierico, Alessandra Rus- so, Sofia Reddel, Giulia Conte, Loris R. Lopetuso, Gianluca Ianiro et al. “Gut microbiota profiling and gut–brain crosstalk in children affected by pediatric acute-onset neuropsychiatric syndrome and pediatric autoimmune neuropsychiatric disor- ders associated with streptococcal infections.” Frontiers in mi- crobiology 9 (2018): 675.
- Murphy, T. K., E. A. Storch, A. Turner, J. M. Reid, J. Tan, and A. B. Lewin. “Maternal history of autoimmune disease in children presenting with tics and/or obsessive–compulsive disorder.” Journal of neuroimmunology 229, no. 1-2 (2010): 243-247.
- Çelik, Gonca Gül, Didem Arslan Taş, Ayşegül Yolga Tahiroglu, Eren Erken, Gülşah Seydaoğlu, Perihan Cam Ray, and Ayşe Avci. “Mannose-binding lectin 2 gene polymorphism in PAN- DAS patients.” Archives of Neuropsychiatry 56, no. 2 (2018): 99.
- Sigra, Sofia, Eva Hesselmark, and Susanne Bejerot. “Treat- ment of PANDAS and PANS: a systematic review.” Neurosci- ence & Biobehavioral Reviews 86 (2018): 51-65.
- Wilbur, Colin, Ari Bitnun, Sefi Kronenberg, Ronald M. Laxer, Deborah M. Levy, William J. Logan, Michelle Shouldice, and E. Ann Yeh. “PANDAS/PANS in childhood: Controversies and evidence.” Paediatrics & child health 24, no. 2 (2019): 85-91.
- Murphy, Tanya K., Wayne K. Goodman, Elia M. Ayoub, and Kytja K. Voeller. “On defining Sydenham’s chorea: where do we draw the line?.” Biological psychiatry 47, no. 10 (2000): 851-857.
- Williams, Kyle A., and Susan E. Swedo. “Post-infectious au- toimmune disorders: Sydenham’s chorea, PANDAS and be- yond.” Brain research 1617 (2015): 144-154.
- Oosterveer, Daniëlla M., Wilhelmina CT Overweg-Plandso- en, and Raymund AC Roos. “Sydenham’s chorea: a practical overview of the current literature.” Pediatric neurology 43, no. 1 (2010): 1-6.
- Boileau, Bernard. “A review of obsessive-compulsive disorder in children and adolescents.” Dialogues in clinical neurosci- ence 13, no. 4 (2011): 401-411.


