Archive : Article / Volume 2, Issue 2
- Case Report | DOI:
- https://doi.org/10.58489/2836-2187/016
Salmonella Cholerasuis an Atypical Presentation and Evolving Drug Resistance
1Department of Microbiology, IGMC Shimla, India
2Department of Orthopaedics, IGMC Shimla, India
3State Epidemiologist, State Institute of Health & Family Welfare, Kasumpti, Shimla, India
4Department of Radiation Diagnosis, IGMC Shimla, India
Pankaj Katoch, Department of Microbiology, IGMC Shimla, India
Pankaj Katoch, et.al. Salmonella Cholerasuis an Atypical Presentation and Evolving Drug Resistance. J. Microb. Res. Vol 2, Iss 2. (2023). DOI: 10.58489/2836-2187/016
© 2023 Pankaj Katoch, this is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Received Date: 29-07-2023
- Accepted Date: 12-08-2023
- Published Date: 22-08-2023
Zoonoses, Salmonella, Non-typhoidal salmonella (NTS), Salmonella cholerasuis, Bacteraemia, Septicaemia, Localized infections
Abstract
Non-typhoidal Salmonella is an important cause of foodborne diseases worldwide. Out of the more than 2500 reported serotypes, Salmonella cholerasuis is believed to be highly invasive and pathogenic to human beings. S. cholerasuis causes a variety of diseases that affect bone, vertebral column, aneurysms, and focal local infections in the human population. The rare occurrence of such an infection in our part of the world, North India, warrants a systematic review of causal factors. This case study aims to document and review the causative factors in the transmission chain and the evolving resistance to antibiotics for a future perspective on this serotype.
Introduction
A certain serotype of Salmonella, that is, the serotype Choleraesuis has a greater tendency to cause bacterial infection in the human population and in swine. This serotype is most commonly isolated from domestic pigs. Domestic pigs and their facilities contaminated by this serotype are some of the risk factors implicated in the transmission of this disease to humans. This zoonotic pathogen, due to its ability to survive for a long time in soil, water, and the environment, remains a constant source of infection. It has also been reported to be isolated from wild boars in Europe [1]. Transmission from wild boars to domestic pigs has been reported in Europe and the United States. Isolates from both wild boar and swine had a very close genetic structure indicating transmission and vice versa [2,3]. The patient had a history of eating swine and wild boar meat, which was significant. We report a rare case of localized osteoarticular infection by Salmonella enterica ssp enterica serovar Cholerasuis that presented in our institution.
Case Report
The study illustrates the isolation of Salmonella, that is, the serotype Cholerasuis, of an immunocompetent patient. The patient had acute osteomyelitis of the calcaneum and had previously undergone an operation for the medial malleolus fracture of the Tibia.
A 54 yrs. an old man who presented on 20 September 2018, with a history of pain in the left heel for the past 10 days that was progressive and sharp and was admitted to the trauma ward of the Orthopedics department. He admitted to having consumed both domestic swine and wild boar meat, which was significant but no other contact with animals or swine was reported.
He had previously been seen in the Outpatient orthopedics department on 13 Sept 2018 and was put on analgesics but the pain persisted and he was admitted on 20 September 2018.
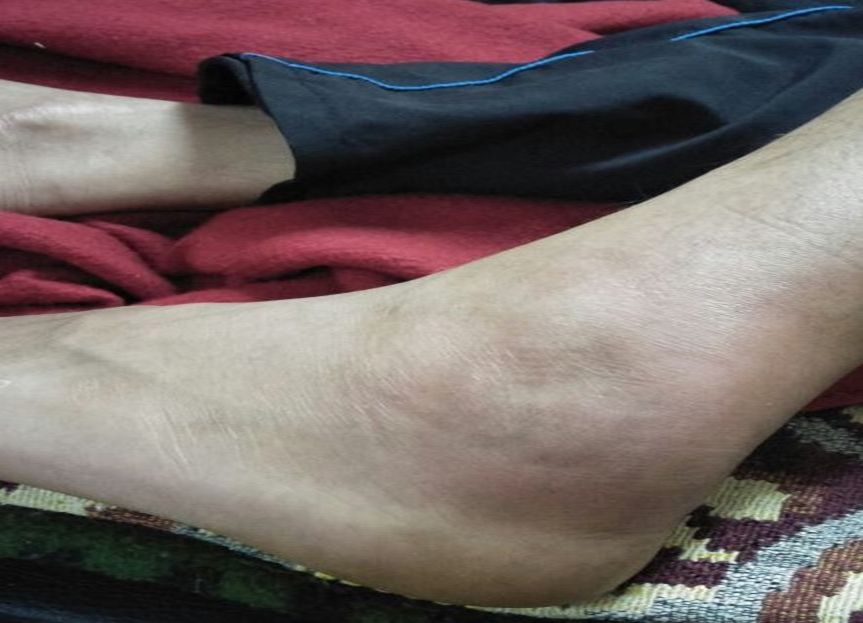
Figure1: Pic of The Affected Part on Presentation to Hospital
He had a history of pyrexia for 3 days, which was of a step ladder pattern. He received Tab Azithromycin 500 mg OD for 3 days and Tab Paracetamol 500 mg, after which the pyrexia subsided. On physical examination, his temp was normal, lung auscultation was normal and no cardiac murmurs were heard. Neurological examination was normal. Blood investigations showed that blood sugar was 128 mg/dl, sodium was 140, potassium 3.6, chloride 99, urea 20, and creatinine 1.1. Hemoglobin 13.6, Red blood cell count 4.63, white blood count 8.70, platelet count 172, hematocrit 41.4%, Mean corpuscular volume 89.4fl, mean corpuscular hemoglobin 29.4 pg., MCHC 32.9, Mean platelet volume10.2, segmented neutrophils 35%, lymphocytes 60%, Absolute lymphocyte count 5.22, C reactive protein (CRP) 143.
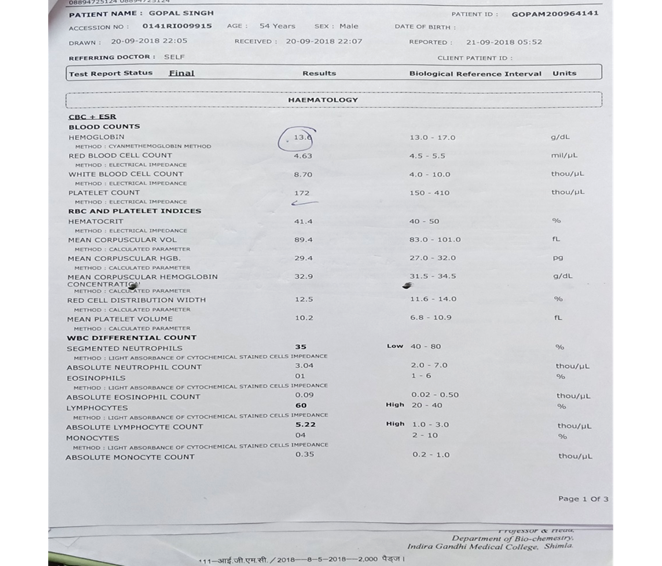
Figure2: Shows Lab Report

Figure3: Shows Lab Report
MRI scan of the patient reported an altered SI involving the left calcaneum showing enhancement with intraosseous abscess and peripherally enhancing collection in the posterior and lateral aspect of the foot and Kagers footpad. Diagnosed as infectious osteomyelitis.

Figure4: SHOWS an MRI Film of the patient showing altered SI involving the left calcaneum showing enhancement with intraosseous abscess
The patient was operated upon bone was drilled and debridement was performed. Figure 8 in Annexure 1 Intraoperative pus was collected and sent for biopsy and culture sensitivity. The patient was empirically placed on injection of cefuroxime and injection of metronidazole based on the sensitivity report (Annexure 2 Table 1). An intraoperative sample of pus was subjected to Ziehl Nelson to rule out tuberculosis. Acid-fast bacilli were not detected on Ziehl Nelson staining ruling out tuberculosis. Giemsa, and Grams staining were also conducted. In Giemsa staining of pus smears, it was reported that it was cellular and showed neutrophils and occasional macrophages. No malignant /atypical cells were seen. The blood culture was sterile. Manual biochemical reactions were put on to confirm the identity of the isolate. IDST Phoenix reported Salmonella enterica ssp enterica serovar Cholerasuis.
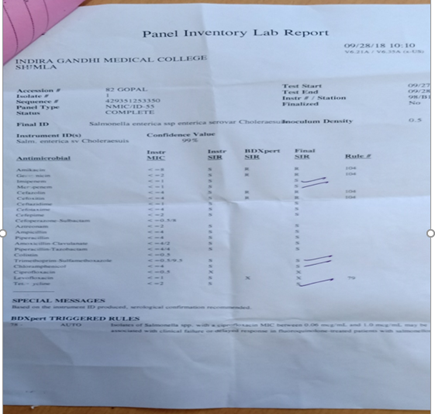
Figure5: An intraoperative sample of pus was inoculated on blood and Mac Conkey agar. Growth on Mac Conkey agar showed non-lactose fermentation colonies.
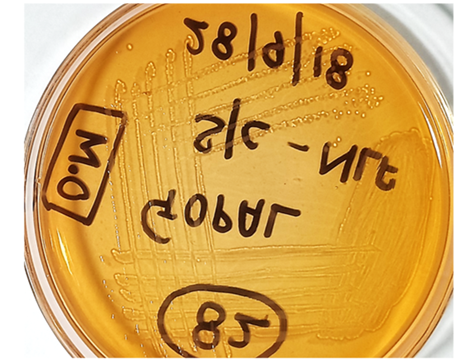
Figure6: Shows Subculture of Non-Lactose Fermenting Colonies from Intra Osseous Pus
Antibiotic Sensitivity of Salmonella Cholerasuis (Annexe-1 Table 1)
| ANTIMICROBIAL | Amk | Gen | Imp | Mer | Cefz | Ctx | Czm | Cpm | CSm | Azt | Amp | Pip | AMC | Ptz | Tmp-smz | Cmpol | Cip | Lev | Tet |
| SENSITIVITY | R | R | S | S | R | R | S | S | S | S | S | S | S | S | S | S | X | X | S |
| MIC | <=8 | <=2 | <=1 | <=1 | <=4 | <=4 | <=1 | <=1 | <=4 | <=2 | <=0.5/8 | <=2 | <=4 | <=4 | <=0.5/9.5 | <=4 | <=0.5 | <=1 | <=2 |
Table1: Antibiotic Sensitivity of Salmonella Cholerasuis
The given isolate was resistant to commonly used antimicrobials like Gentamicin, Amikacin, Cefazolin, Ciprofloxacin, Levofloxacin, and Cefoxitin and was sensitive to Ampicillin, Piperacillin, Ceftazidime, Cefotaxime, combinations of Amoxicillin Clavulanate, Piperacillin Tazobactum, Trimethoprim-Sulfamethoxazole, Carbapenems like Meropenem, Imipenem, and Aztreonam.
Discussion
Most cases of Salmonella species infection are due to contaminated food and water items and is one of the most important causes of death, accounting for nearly 30% of deaths due to foodborne diseases in the United States while data from other countries are not available [4]. It causes a huge financial burden on economies all over the world. Salmonella infection can also be transmitted by personal contact, nosocomial transmission, animals, and contaminated drugs. Cooked undercooked meat of pigs is considered to be the main reason for infection by Salmonella Cholerasuis [5]. This infection serotype can rapidly lead to bacteremia with little or no intestinal involvement [6]. A study in Taiwan reported that serotype Choleraesuis is the most invasive pointing to the fact that the highest number of extraintestinal infections are caused by it [7]. A study reported that the highest incidence of septicemia was due to infections with Choleraesuis and Dublin serotype.
There has been a rapid increase in the number of reported cases of Salmonella cholerasuis. Salmonellae species cause a variety of human diseases ranging from gastroenteritis, typhoid fever, bacteremia, and even an asymptomatic state [9]. Nontyphoidal Salmonella bacteremia can lead to serious infections in the patient who suffers from comorbidity leading to the development of localized infections like septic arthritis and osteomyelitis. It has been pointed out before that serotype Choleraesuis may be transmitted to any site in the body by a hematogenous route leading to local infection, even though the bacterial infection is successfully treated [10,11]. A very important question that needs to be addressed is how similar bacteria, of the same subspecies S. enterica ssp, cause different clinical presentations in humans despite more than 95% shared genomic sequence [12].
It can be presumed due to the acquisition of pathogenic traits by horizontal transmission. Some studies opined that NTS serovars might have evolved some molecular and genetic ways to overcome gut microflora. It is submitted that Typhoidal strains are not reported to possess this characteristic and grow in blood [13]. There is a lot of difference in humans when comparing Salmonella and non-typhoidal Salmonella infection-induced cytokine levels [14]. Serotype Choleraesuis may have unique invasion genes for cell infectivity [15].
We need to understand the genetic mechanisms responsible for the differences in disease presentation and prognosis through research on animal models. The most consistent systemic lesion of S. choleraesuis infection was interstitial pneumonia and multifocal hepatic necrosis in pigs challenged with this infection. Certain strains of swine are thought to be a causative factor in contributing to multidrug resistance mutations such as (Gyr-Aand ParC) and genes such as (CMY-2 and AMP-C) leading to the acquisition of extended-spectrum beta-lactam resistance. The virulence plasmid is considered to be crucial and other gene-mediated adhesions contribute to its increased pathogenesis and virulence [16]. Integrons have a decisive role in the acquisition and dissemination of qac genes in salmonella species [17]. Acute osteomyelitis most frequently manifests in children whereas adults are generally less affected and Staphylococcus aureus is most frequently implicated.
Post-traumatic osteomyelitis can occur after an uneventful postoperative period, where the dormant infection is reactivated again. The most important presentation is localized pain. Systemic involvement features such as fever and chills due to inflammation are minimal compared to acute bacteraemia patients. We are not able to presume the factors due to which pathogenic bacteria become reactivated from a dormant state and cause acute clinical signs and symptoms, and what factors lead to their reactivation. CRP and ESR generally show elevated levels. The importance of a high degree of suspicion despite negative blood cultures, cultures, X-rays, CT scans, and MRIs cannot be overemphasized. Patients suffering from sickle-cell anemia osteomyelitis and joint infections are difficult to treat, as antibiotic resistance has been reported [18]. After intestinal salmonellosis infection, a complication that is prolonged polyarticular reactive joint can occur and not respond to antibiotic therapy. The role of culture sensitivity after surgery and appropriate antibiotic selection for the full duration is essential to prevent relapse. S. cholerasuis bacteremia usually leads to extra-intestinal infections, particularly in risk groups that are people exposed to trauma or surgery, elderly with age-related complications, and patients on immunosuppressive drugs.
Conclusions
The animals used for food production in contaminated containment facilities and slaughterhouses are a constant source of infection. The causative factor is Zoonotic in this case and is of public health importance. Salmonella serotypes indicate the emergence of antibiotic resistance. There should be strict control on antibiotic usage in animals to minimize the risk of transmission of antibiotic-resistant serotypes to humans.
References
- Mead, Paul S., Laurence Slutsker, Vance Dietz, Linda F. McCaig, Joseph S. Bresee, Craig Shapiro, Patricia M. Griffin, and Robert V. Tauxe. “Food-related illness and death in the United States.” Emerging infectious diseases 5, no. 5 (1999): 607.
- Chen, Yen-Hsu, Tyen-Po Chen, Po-Lian Lu, Yu-Chieh Su, Kao-Pin Hwang, Jih-Jin Tsai, Hsiao-Hsu Cheng, and Chien-Fang Peng. “Salmonella choleraesuis bacteremia in southern Taiwan.” The Kaohsiung journal of medical sciences 15, no. 4 (1999): 202-208.
- Shimoni, Zvi, Silvio Pitlik, Leonard Leibovici, Zmira Samra, Hanna Konigsberger, Moshe Drucker, Vered Agmon, Shai Ashkenazi, and Miriam Weinberger. “Nontyphoid Salmonella bacteremia: age-related differences in clinical presentation, bacteriology, and outcome.” Clinical infectious diseases 28, no. 4 (1999): 822-827.
- Chiu, Cheng-Hsun, Tzou-Yien Lin, and Jonathan T. Ou. “Prevalence of the virulence plasmids of nontyphoid Salmonella in the serovars isolated from humans and their association with bacteremia.” Microbiology and immunology 43, no. 9 (1999): 899-903.
- Threlfall, E. J., M. L. Hall, and B. Rowe. “Salmonella bacteraemia in England and Wales, 1981-1990.” Journal of Clinical Pathology 45, no. 1 (1992): 34-36.
- Steinebrunner, Niels, Catharina Sandig, Stefan Zimmermann, Wolfgang Stremmel, Christoph Eisenbach, and Alexander Mischnik. “Salmonella enterica serovar Minnesota urosepsis in a patient with Crohn’s disease in the absence of recent or current gastrointestinal symptoms.” Journal of medical microbiology 62, no. 9 (2013): 1360-1362.
- Reed, Willie M., Harvey J. Olander, and H. Leon Thacker. “Studies on the pathogenesis of Salmonella typhimurium and Salmonella choleraesuis var kunzendorf infection in weanling pigs.” American journal of veterinary research 47, no. 1 (1986): 75-83.
- Pospischil, Andreas, Richard L. Wood, and Timothy D. Anderson. “Peroxidase-antiperoxidase and immunogold labeling of Salmonella typhimurium and Salmonella choleraesuis var kunzendorf in tissues of experimentally infected swine.” American Journal of Veterinary Research 51, no. 4 (1990): 619-624.
- Kelly, Sandra M., BETH A. Bosecker, and R. Curtiss 3rd. “Characterization and protective properties of attenuated mutants of Salmonella choleraesuis.” Infection and immunity 60, no. 11 (1992): 4881-4890.
- Santos, Edwin M., and Francisco L. Sapico. “Vertebral osteomyelitis due to salmonellae: report of two cases and review.” Clinical Infectious Diseases 27, no. 2 (1998): 287-295.
- Bai, Li, Ruiting Lan, Xiuli Zhang, Shenghui Cui, Jin Xu, Yunchang Guo, Fengqin Li, and Ding Zhang. “Prevalence of Salmonella isolates from chicken and pig slaughterhouses and emergence of ciprofloxacin and cefotaxime co-resistant S. enterica serovar Indiana in Henan, China.” PloS one 10, no. 12 (2015): e0144532.
- Alpuche Aranda, Celia M., Joel A. Swanson, WENDY P. LooMIs, and Samuel I. Miller. “Salmonella typhimurium activates virulence gene transcription within acidified macrophage phagosomes.” Proceedings of the National Academy of Sciences 89, no. 21 (1992): 10079-10083.
- Jean, Shio-Shin, Jen-Yu Wang, and Po-Ren Hsueh. “Bacteremia caused by Salmonella enterica serotype Choleraesuis in Taiwan.” Journal of microbiology, immunology, and infection= Wei mian yu gan ran za zhi 39, no. 5 (2006): 358-365.
- COHEN, JEFFREY I., John A. Bartlett, and G. Ralph Corey. “Extra-intestinal manifestations of Salmonella infections.” Medicine 66, no. 5 (1987): 349-388.
- COHEN, JEFFREY I., John A. Bartlett, and G. Ralph Corey. “Extra-intestinal manifestations of Salmonella infections.” Medicine 66, no. 5 (1987): 349-388.


